Drug Details
| General Information of the Drug (ID: DR0067) | ||||
|---|---|---|---|---|
| Name |
3-methyladenine
|
|||
| Synonyms |
3-methyladenine; 5142-23-4; 3-Methyl-3H-purin-6-amine; 6-Amino-3-methylpurine; 3-MA; 3-Methyl-3H-adenine; 3H-Purin-6-amine, 3-methyl-; NSC 66389; n3-methyladenine; ADENINE, 3-METHYL-; MFCD00010531; CHEBI:38635; UNII-DR88TV7SNU; DR88TV7SNU; 3-METHYL-3H-PURIN-6-YLAMINE; 3-Methyladenine autophagy inhibitor; 3-Methyladenine, 90+%; 3-MA nucleobase; 3-Methyladenine (3-MA); 3MA; 60192-57-6; N(3)-methyladenine; 3-methylpurin-6-amine; EINECS 225-908-6; BRN 0146087; 3-methyl-7H-purin-6-imine; 3mea; 3-methyl-Adenine; 3-Methyladenine?; NSC66389; 1p7m; 4ai5; Autophagy Inhibitor, 3-MA; NCIOpen2_000270; SCHEMBL48369; 5-26-17-00151 (Beilstein Handbook Reference); MLS006010995; SCHEMBL254058; CHEMBL292268; 3-methyl-3-hydropurin-6-imine; CHEMBL4303725; SCHEMBL15764619; 3-Methyl-3H-purin-6-amine #; AOB2548; DTXSID80199406; EX-A130; 3-methyl-3-hydropurine-6-ylamine; Bio1_000422; Bio1_000911; Bio1_001400; HMS3656P04; ZINC403086; 3-Methyl-7H-purine-6(3H)-imine; ACN-C001131; AMY11868; BCP02452; 3-Methyladenine, autophagy inhibitor; ABP000334; ANW-54285; BDBM50488841; NSC-66389; QC-518; s2767; SBB086626; AKOS003382321; AKOS006228458; ZINC100003619; ACN-001131; CCG-206388; CS-5207; DB04104; MCULE-5086156902; NSC 66389 (3-MA); VZ27096; NCGC00345447-02; AC-28818; AK-94088; AS-19224; HY-19312; SMR002530641; SY026559; AB0007938; DB-051961; A7582; FT-0635560; FT-0671432; M2518; ST50298916; SW220216-1; X5709; 3-methyl-3H-purin-6-amine (ACD/Name 4.0); C00913; Q-9331; 3-methyl-3H-purin-6-ylamine (ACD/Name 4.0); J-640198; J-800199; W-202935; BRD-K81647657-001-01-9; Q27094948
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Cardiac injury [ICD-11: NB31] | Investigative | [1] | |
| Structure |
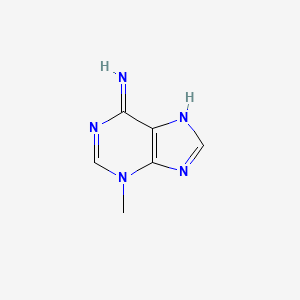
|
Click to Download Mol2D MOL |
||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C6H7N5
|
|||
| PubChem CID | ||||
| Canonical SMILES |
CN1C=NC(=N)C2=C1N=CN2
|
|||
| InChI |
1S/C6H7N5/c1-11-3-10-5(7)4-6(11)9-2-8-4/h2-3,7H,1H3,(H,8,9)
|
|||
| InChIKey |
ZPBYVFQJHWLTFB-UHFFFAOYSA-N
|
|||
| CAS Number |
CAS 5142-23-4
|
|||
| ChEBI ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Pterostilbene | Vitis vinifera | Click to Show/Hide the Molecular Data of This NP | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Down-regulation | Expression | VIM | Molecule Info |
Pathway MAP
|
|
| In-vitro Model | MDA-MB-231 | CVCL_0062 | Breast adenocarcinoma | Homo sapiens | ||
| BT-549 | CVCL_1092 | Invasive breast carcinoma | Homo sapiens | |||
| Experimental
Result(s) |
The anti-tumor efficiency of pterostilbene is promoted with a combined treatment of Fas signaling or autophagy inhibitors in triple negative breast cancer cells. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Arachidonate 5-lipoxygenase (5-LOX) | Molecule Info | [3] | |
| BioCyc | Aspirin-triggered lipoxin biosynthesis | Click to Show/Hide | ||
| 2 | Resolvin D biosynthesis | |||
| 3 | Leukotriene biosynthesis | |||
| 4 | Lipoxin biosynthesis | |||
| 5 | Aspirin triggered resolvin D biosynthesis | |||
| 6 | Aspirin triggered resolvin E biosynthesis | |||
| KEGG Pathway | Arachidonic acid metabolism | Click to Show/Hide | ||
| 2 | Metabolic pathways | |||
| 3 | Serotonergic synapse | |||
| 4 | Ovarian steroidogenesis | |||
| 5 | Toxoplasmosis | |||
| NetPath Pathway | IL4 Signaling Pathway | Click to Show/Hide | ||
| Pathwhiz Pathway | Arachidonic Acid Metabolism | Click to Show/Hide | ||
| WikiPathways | Vitamin D Receptor Pathway | Click to Show/Hide | ||
| 2 | Arachidonic acid metabolism | |||
| 3 | Eicosanoid Synthesis | |||
| 4 | Selenium Micronutrient Network | |||