Drug Details
| General Information of the Drug (ID: DR4974) | ||||
|---|---|---|---|---|
| Name |
Ketoconazole
|
|||
| Synonyms |
ketoconazole; 65277-42-1; (+)-Ketoconazole; (2R,4S)-ketoconazole; Kuric; Ketocanazole; CPD000058460; CHEMBL75; MFCD00058579; C26H28Cl2N4O4; SMR000058460; MLS000069784; MLS001146934; 1-[4-[4-[[(2R,4S)-2-(2,4-dichlorophenyl)-2-(imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]ethanone; 1-acetyl-4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy}phenyl)piperazine; 142128-59-4; CHEBI:48336; DSSTox_CID_9879; DSSTox_RID_78829; DSSTox_GSID_29879; 1-(4-(4-(((2R,4S)-2-((1H-IMIDAZOL-1-YL)METHYL)-2-(2,4-DICHLOROPHENYL)-1,3-DIOXOLAN-4-YL)METHOXY)PHENYL)PIPERAZIN-1-YL)ETHANONE; R-41400; rac-trans-Ketoconazole; KTZ; SR-01000075626; SR-01000597381; Ketoconazole (k); Ketoconazole,(S); NCGC00016907-01; 83374-59-8; Prestwick_744; CAS-65277-42-1; Tocris-1103; Opera_ID_397; Prestwick0_000389; Prestwick1_000389; Prestwick2_000389; Prestwick3_000389; R 41,400; UPCMLD-DP138; SCHEMBL8407; Lopac0_000666; BSPBio_000577; MLS000758224; MLS001423987; MLS002207053; MLS002222255; BIDD:GT0696; US9150527, Ketoconazole; SPBio_002498; AMY917; BDBM8610; BPBio1_000635; DTXSID7029879; UPCMLD-DP138:001; BDBM60666; HY-B0105A; Ketoconazole, >=98% (HPLC); HMS1569M19; HMS2051A19; HMS2089N05; HMS2096M19; HMS2234H17; HMS3262E13; HMS3414J19; HMS3678J17; HMS3713M19; ZINC643138; BCP28528; Piperazine, (+/-)-1-acetyl-4-[4-[[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-, rel-; Ketoconazole 2.0 mg/ml in Methanol; Tox21_110676; Tox21_300267; Tox21_500666; EI-107; s1353; AKOS007930650; AB02344; CCG-100815; CS-1846; KS-1205; LP00666; MCULE-2726394491; NC00065; SDCCGSBI-0050645.P002; MRF-0000100; 24F2-1,25(OH)D3; NCGC00025000-01; NCGC00025000-02; NCGC00025000-03; NCGC00025000-04; NCGC00025000-05; NCGC00025000-06; NCGC00025000-07; NCGC00025000-08; NCGC00025000-09; NCGC00025000-10; NCGC00025000-14; NCGC00025000-16; NCGC00025000-28; NCGC00253967-01; NCGC00261351-01; (+/-)-cis-1-Acetyl-4-[4-[[2-(2,4-dichlorophenyl)-2-(imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazine; AC-15957; cis-1-Acetyl-4-[4-[[2-(2,4-; AB0090681; Ketoconazole 100 microg/mL in Acetonitrile; EU-0100666; K0045; SW196888-4; BIM-0050645.0001; J10202; K 1003; W-2941; 277K421; Ketoconazole, Antibiotic for Culture Media Use Only; Q-201267; SR-01000075626-1; SR-01000075626-4; SR-01000597381-1; SR-01000597381-6; BRD-K29113274-001-03-6; BRD-K29113274-001-11-9; BRD-K29113274-001-21-8; Q27121163; Ketoconazole, British Pharmacopoeia (BP) Reference Standard; UNII-R9400W927I component XMAYWYJOQHXEEK-OZXSUGGESA-N; Ketoconazole, European Pharmacopoeia (EP) Reference Standard; Ketoconazole, United States Pharmacopeia (USP) Reference Standard; dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-piperazine; Ketoconazole, Pharmaceutical Secondary Standard; Certified Reference Material; (+/-)-cis-1-Acetyl-4-(4-[(2-[2,4-dichlorophenyl]-2-[1H-imidazol-1-ylmethyl]-1,3-dioxolan-4-yl)-methoxy]phenyl)piperazine; (2R,4S)-1-acetyl-4-(4-{[2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy}phenyl)piperazine; 1-(4-(4-(((2R,4S)-2-((1H-imidazol-1-yl)methyl)-2-(2,4-dichlorophenyl)-1,3-dioxolan-4-yl)methoxy)phenyl)piperazin-1-yl)ethan-1-one; 1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy}phenyl)piperazin-1-yl]ethan-1-one; cis-1-Acetyl-4-[4-[[2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl] methoxy]phenyl]piperazine; Ethanone, 1-[4-[4-[[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-1-piperazinyl]-; KZ; Piperazine, (+)-1-acetyl-4-[4-[[2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Fungal infection [ICD-11: 1F29-1F2F] | Approved | [1] | |
| Structure |
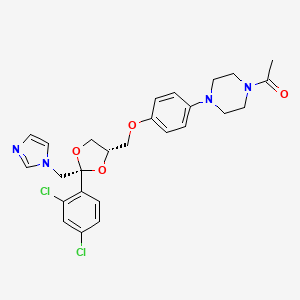
|
Click to Download Mol2D MOL |
||
| ADMET Property |
Absorption Cmax
The maximum plasma concentration (Cmax) of drug is 2.5-3 mg/L
Absorption Tmax
The time to maximum plasma concentration (Tmax) is 1-4 h
BDDCS Class
Biopharmaceutics Drug Disposition Classification System (BDDCS) Class 2: low solubility and high permeability
Bioavailability
75% of drug becomes completely available to its intended biological destination(s)
Clearance
The clearance of drug is 8.66 L/h
Elimination
Only 2-4% of the ketoconazole dose is eliminated unchanged in the urine, and over 95% is eliminated through hepatic metabolism
Half-life
The concentration or amount of drug in body reduced by one-half in 2 hours
MRTD
The Maximum Recommended Therapeutic Dose (MRTD) of drug that ensured maximising efficacy and moderate side effect is 10.7525 micromolar/kg/day
Vd
The volume of distribution (Vd) of drug is 25.41 L or 0.36 L/kg
Water Solubility
The ability of drug to dissolve in water is measured as 0.0069 mg/mL
Click to Show/Hide
|
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C26H28Cl2N4O4
|
|||
| PubChem CID | ||||
| Canonical SMILES |
CC(=O)N1CCN(CC1)C2=CC=C(C=C2)OCC3COC(O3)(CN4C=CN=C4)C5=C(C=C(C=C5)Cl)Cl
|
|||
| InChI |
1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m0/s1
|
|||
| InChIKey |
XMAYWYJOQHXEEK-OZXSUGGESA-N
|
|||
| CAS Number |
CAS 65277-42-1
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Xanthotoxin | Cullen corylifolium | Click to Show/Hide the Molecular Data of This NP | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Candida albicans ATCC 22972 | Microorganism model | Candida albicans | |||
| Candida glabrata ATCC 90525 | Microorganism model | Candida glabrata | ||||
| Candida guilliermondii ATCC 20216 | Microorganism model | Candida guilliermondii | ||||
| Candida krusei ATCC 6258 | Microorganism model | Candida krusei | ||||
| Candida parapsilosis ATCC 7330 | Microorganism model | Candida parapsilosis | ||||
| Candida tropicalis ATCC 42678 | Microorganism model | Candida tropicalis | ||||
| Experimental
Result(s) |
1/2 MIC dose of xanthorrhizol in combination with 1/2 MIC dose of ketoconazole or 1/2 MIC dose of amphotericin B exhibited growth inhibition of all Candida species tested and reduced viable cells by several logs within 4 h. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Candida Cytochrome P450 51 (Candi ERG11) | Molecule Info | [3] | |

