Drug Details
| General Information of the Drug (ID: DR5683) | ||||
|---|---|---|---|---|
| Name |
Montelukast
|
|||
| Synonyms |
montelukast; 158966-92-8; Singulair; UNII-MHM278SD3E; (R-(E))-1-(((1-(3-(2-(7-Chloro-2-quinolinyl)ethenyl)phenyl)-3-(2-(1-hydroxy-1-methylethyl)phenyl)propyl)thio)methyl)cyclopropaneacetic acid; CHEMBL787; MHM278SD3E; CHEBI:50730; Cyclopropaneacetic acid, 1-((((1R)-1-(3-((1E)-2-(7-chloro-2-quinolinyl)ethenyl)phenyl)-3-(2-(1-hydroxy-1-methylethyl)phenyl)propyl)thio)methyl)-; Montelukast [INN:BAN]; (R,E)-2-(1-(((1-(3-(2-(7-chloroquinolin-2-yl)vinyl)phenyl)-3-(2-(2-hydroxypropan-2-yl)phenyl)propyl)thio)methyl)cyclopropyl)acetic acid; 142522-28-9; Aerokast; Brondilat (TN); 1-[[[(1 R)-1-[3-[(1E)-2-(7-chloro-2-quinolinyl)ethenyl] phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropaneacetic acid; 2-[1-({[(1R)-1-{3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl}-3-[2-(2-hydroxypropan-2-yl)phenyl]propyl]sulfanyl}methyl)cyclopropyl]acetic acid; Montelukast (INN); SR-01000763441; HSDB 7582; 1-((((1R)-1-(3-((1E)-2-(7-Chloro-2-quinolinyl)ethenyl)phenyl)-3-(2-(1-hydroxy-1-methylethyl)phenyl)propyl)thio)methyl)cyclopropaneacetic acid; cc-91; SCHEMBL4486; SCHEMBL4487; {1-[({(1R)-1-{3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl}-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl}sulfanyl)methyl]cyclopropyl}acetic acid; BIDD:GT0394; GTPL3340; DTXSID9023334; CHEBI:94710; HMS2089D07; HMS3715N13; HMS3886L03; ACT04773; ZINC3831151; BDBM50052024; HY-13315A; AKOS025310659; CCG-221186; DB00471; SB19081; NCGC00188977-01; {1-[({(1R)-1-{3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl}-3-[2-(2-hydroxypropan-2-yl)phenyl]propyl}sulfanyl)methyl]cyclopropyl}acetic acid; 2-[1-[[(1R)-1-[3-[(E)-2-(7-chloro-2-quinolyl)vinyl]phenyl]-3-[2-(1-hydroxy-1-methyl-ethyl)phenyl]propyl]sulfanylmethyl]cyclopropyl]acetic acid; Cyclopropaneacetic acid, 1-(((1-(3-(2-(7-chloro-2-quinolinyl)ethenyl)phenyl)-3-(2-(1-hydroxy-1-methylethyl)phenyl)propyl)thio)methyl)-, (R-(E))-; CS-0002595; S5783; 5306-EP2270008A1; 5306-EP2276739A1; 5306-EP2280006A1; 5306-EP2281813A1; 5306-EP2281815A1; 5306-EP2281819A1; 5306-EP2284166A1; 5306-EP2287154A1; 5306-EP2288595A2; 5306-EP2292617A1; 5306-EP2292619A1; 5306-EP2295409A1; 5306-EP2298415A1; 5306-EP2301933A1; 5306-EP2305640A2; 5306-EP2305659A1; 5306-EP2308562A2; 5306-EP2311818A1; 5306-EP2311827A1; 5306-EP2314590A1; C07482; D08229; AB01275454-01; 522M289; Q417767; SR-01000763441-3; SR-01000763441-5; BRD-K99673372-001-01-9; 1-[[[(R)-3-[2-(1-Hydroxy-1-methylethyl)phenyl]-1-[3-[2-(7-chloroquinoline-2-yl)ethenyl]phenyl]propyl]thio]methyl]cyclopropane-1-acetic acid; 1124196-02-6; r-(e)-1-[[[1-[3-[2-(7-chloro-2-quinolinyl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]thio]methyl]cyclopropaneacetic acid
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Asthma [ICD-11: CA23] | Approved | [1] | |
| Structure |
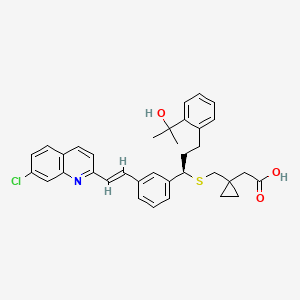
|
Click to Download Mol2D MOL |
||
| ADMET Property |
Absorption AUC
The area under the plot of plasma concentration (AUC) of drug is 642870000 mgh/L
Absorption Tmax
The time to maximum plasma concentration (Tmax) is 2-4 h
BDDCS Class
Biopharmaceutics Drug Disposition Classification System (BDDCS) Class 2: low solubility and high permeability
Bioavailability
The bioavailability of drug is 90%
Bioavailability
62% of drug becomes completely available to its intended biological destination(s)
Clearance
The clearance of drug is 45 mL/min
Clearance
The drug present in the plasma can be removed from the body at the rate of 0.68 mL/min/kg
Elimination
0.2% of drug is excreted from urine in the unchanged form
Half-life
The concentration or amount of drug in body reduced by one-half in 2.7 - 5.5 hours
Half-life
The concentration or amount of drug in body reduced by one-half in 5 hours
Metabolism
The drug is metabolized via the cytochrome P450 3A4, 2C8, and 2C9 isoenzymes
MRTD
The Maximum Recommended Therapeutic Dose (MRTD) of drug that ensured maximising efficacy and moderate side effect is 0.28489 micromolar/kg/day
Unbound Fraction
The unbound fraction of drug in plasma is 0.002%
Vd
The volume of distribution (Vd) of drug is 8-11 L
Vd
Fluid volume that would be required to contain the amount of drug present in the body at the same concentration as in the plasma 0.15 L/kg
Click to Show/Hide
|
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C35H36ClNO3S
|
|||
| PubChem CID | ||||
| Canonical SMILES |
CC(C)(C1=CC=CC=C1CCC(C2=CC=CC(=C2)C=CC3=NC4=C(C=CC(=C4)Cl)C=C3)SCC5(CC5)CC(=O)O)O
|
|||
| InChI |
1S/C35H36ClNO3S/c1-34(2,40)30-9-4-3-7-25(30)13-17-32(41-23-35(18-19-35)22-33(38)39)27-8-5-6-24(20-27)10-15-29-16-12-26-11-14-28(36)21-31(26)37-29/h3-12,14-16,20-21,32,40H,13,17-19,22-23H2,1-2H3,(H,38,39)/b15-10+/t32-/m1/s1
|
|||
| InChIKey |
UCHDWCPVSPXUMX-TZIWLTJVSA-N
|
|||
| CAS Number |
CAS 158966-92-8
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Iloprost | Homo sapiens | Click to Show/Hide the Molecular Data of This NP | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Down-regulation | Expression | IL6 | Molecule Info |
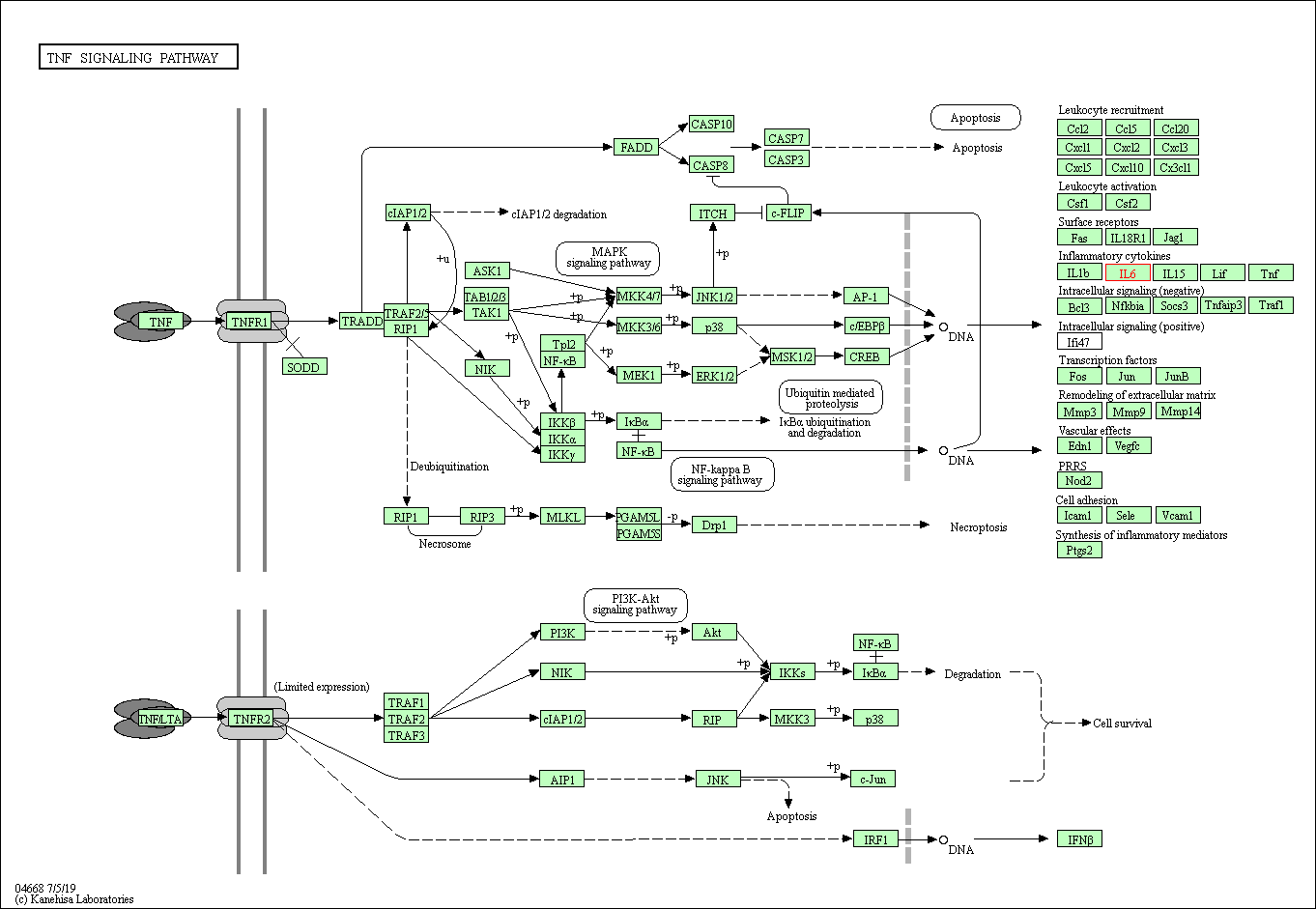
Pathway MAP
|
|
| Experimental
Result(s) |
Combined use of iloprost and montelukast may reduce ischemic damage in transient spinal cord ischemia and may provide better neurological outcome. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Leukotriene CysLT1 receptor (CYSLTR1) | Molecule Info | [3] | |
| KEGG Pathway | Calcium signaling pathway | Click to Show/Hide | ||
| 2 | Neuroactive ligand-receptor interaction | |||
| NetPath Pathway | TGF_beta_Receptor Signaling Pathway | Click to Show/Hide | ||
| 2 | IL4 Signaling Pathway | |||
| 3 | IL3 Signaling Pathway | |||
| Pathway Interaction Database | Endothelins | Click to Show/Hide | ||
| Reactome | Leukotriene receptors | Click to Show/Hide | ||
| 2 | G alpha (q) signalling events | |||
| WikiPathways | GPCRs, Class A Rhodopsin-like | Click to Show/Hide | ||
| 2 | Gastrin-CREB signalling pathway via PKC and MAPK | |||
| 3 | GPCR ligand binding | |||
| 4 | GPCR downstream signaling | |||