Natural Product (NP) Details
| General Information of the NP (ID: NP1844) | |||||
|---|---|---|---|---|---|
| Name |
Berberine
|
||||
| Synonyms |
Berberin; Umbellatine; UNII-0I8Y3P32UF; 0I8Y3P32UF; CHEBI:16118; EINECS 218-229-1; Berberal; BRN 3570374; ST055798; 9,10-Dimethoxy-2,3-(methylenedioxy)-7,8,13,13a-tetrahydroberbinium; Benzo(g)-1,3-benzodioxolo(5,6-a)quinolizinium, 5,6-dihydro-9,10-dimethoxy-; 9,10-dimethoxy-5,6-dihydro[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinolin-7-ium; Berbamine sulphate acid; CHEMBL12089; 7,8,13,13a-tetradehydro-9,10-dimethoxy-2,3-(methylenedioxy)berbinium; BERBINIUM, 7,8,13,13a-TETRAHYDRO-9,10-DIMETHOXY-2,3-(METHYLE
Click to Show/Hide
|
||||
| Species Origin | Astragalus membranaceus ... | Click to Show/Hide | |||
| Astragalus membranaceus | |||||
| Berberis poiretii | |||||
| Disease | Colorectal cancer [ICD-11: 2B91] | Phase 3 | [1] | ||
| Structure |
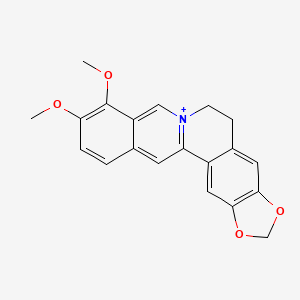
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-5.01
MDCK Permeability
-4.7
PAMPA
- -
HIA
- - -
Distribution
VDss
0.073
PPB
68.4%
BBB
- -
Metabolism
CYP1A2 inhibitor
++
CYP1A2 substrate
+++
CYP2C19 inhibitor
- - -
CYP2C19 substrate
-
CYP2C9 inhibitor
- - -
CYP2C9 substrate
++
CYP2D6 inhibitor
+++
CYP2D6 substrate
+++
CYP3A4 inhibitor
+++
CYP3A4 substrate
- - -
CYP2B6 inhibitor
+++
CYP2B6 substrate
+++
CYP2C8 inhibitor
- - -
HLM Stability
+
Excretion
CLplasma
5.282
T1/2
1.288
Toxicity
DILI
- -
Rat Oral Acute Toxicity
++
FDAMDD
+++
Respiratory
+++
Human Hepatotoxicity
- -
Ototoxicity
- - -
Drug-induced Nephrotoxicity
- -
Drug-induced Neurotoxicity
- - -
Hematotoxicity
- - -
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C20H18NO4+
|
||||
| PubChem CID | |||||
| Canonical SMILES |
COC1=C(C2=C[N+]3=C(C=C2C=C1)C4=CC5=C(C=C4CC3)OCO5)OC
|
||||
| InChI |
1S/C20H18NO4/c1-22-17-4-3-12-7-16-14-9-19-18(24-11-25-19)8-13(14)5-6-21(16)10-15(12)20(17)23-2/h3-4,7-10H,5-6,11H2,1-2H3/q+1
|
||||
| InChIKey |
YBHILYKTIRIUTE-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 2086-83-1
|
||||
| ChEBI ID | |||||
| Herb ID | |||||
| ETMC ID | |||||
| SymMap ID | |||||
| TCMSP ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Cisplatin | Bladder cancer | Click to Show/Hide the Molecular Data of This Drug | ||||
| Augmenting Drug Sensitivity | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
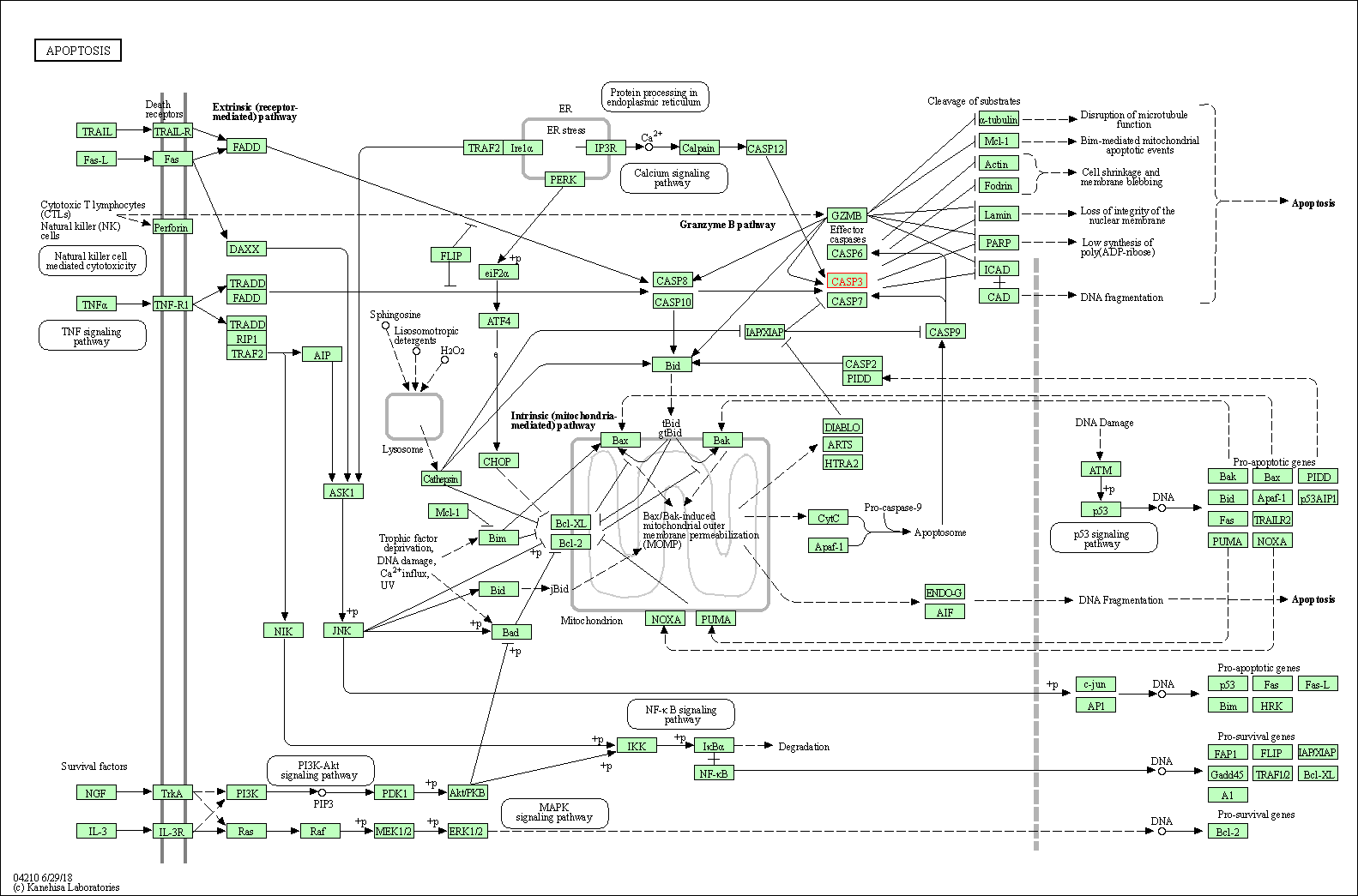
Up-regulation | Cleavage | CASP3 | Molecule Info |
Pathway MAP
|
|
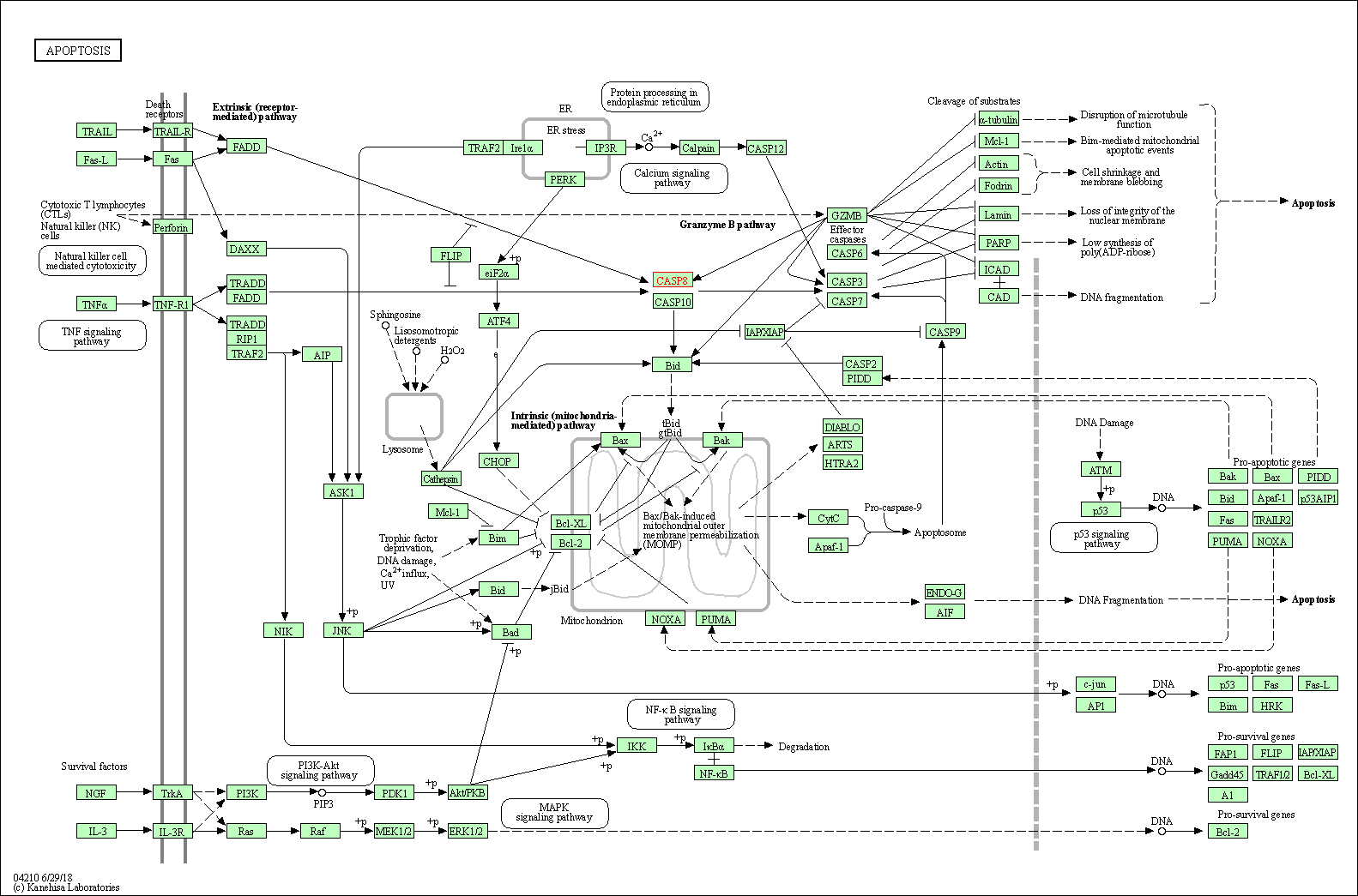
| Up-regulation | Cleavage | CASP8 | Molecule Info |
Pathway MAP
|
||
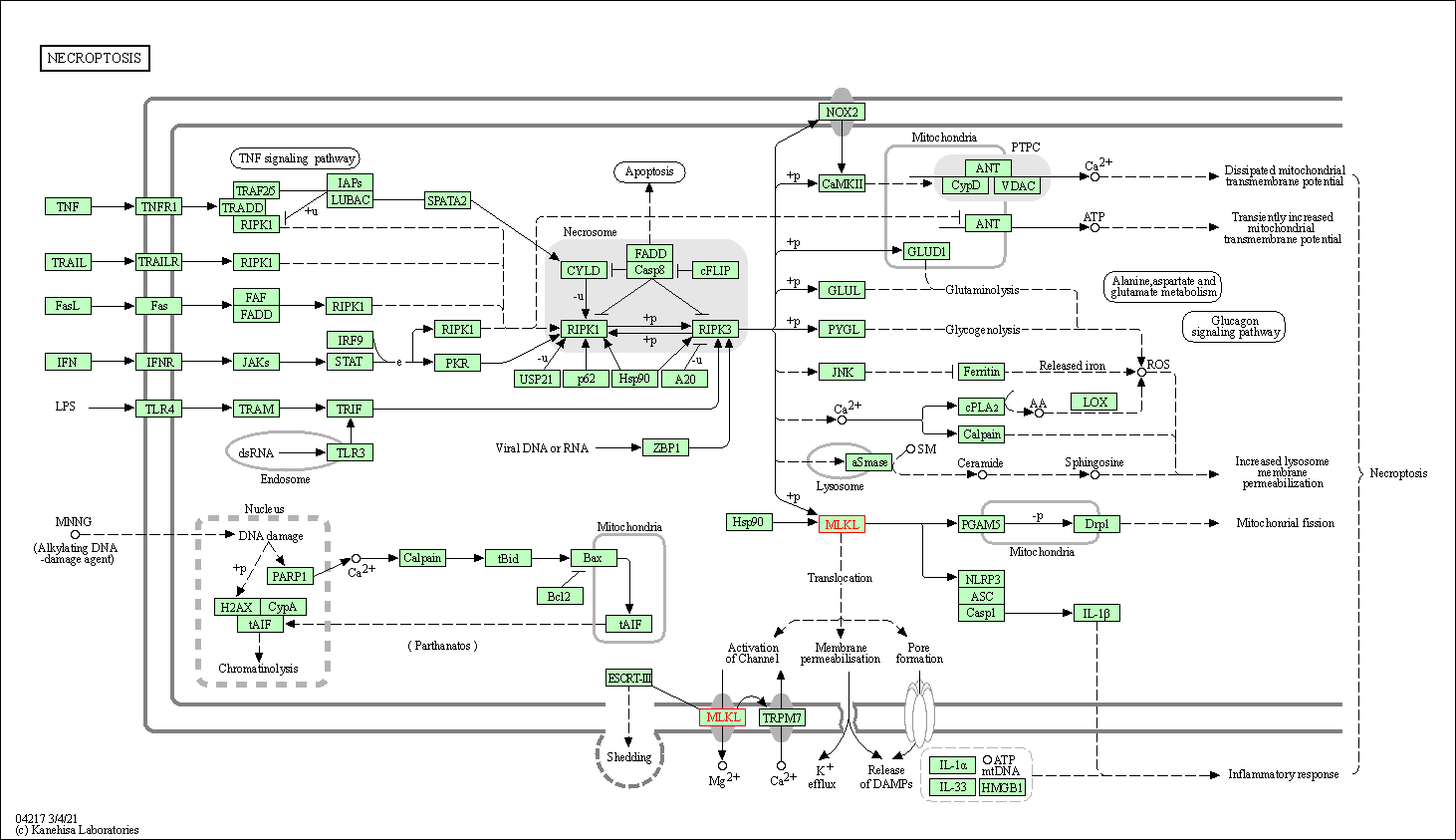
| Up-regulation | Phosphorylation | MLKL | Molecule Info |
Pathway MAP
|
||
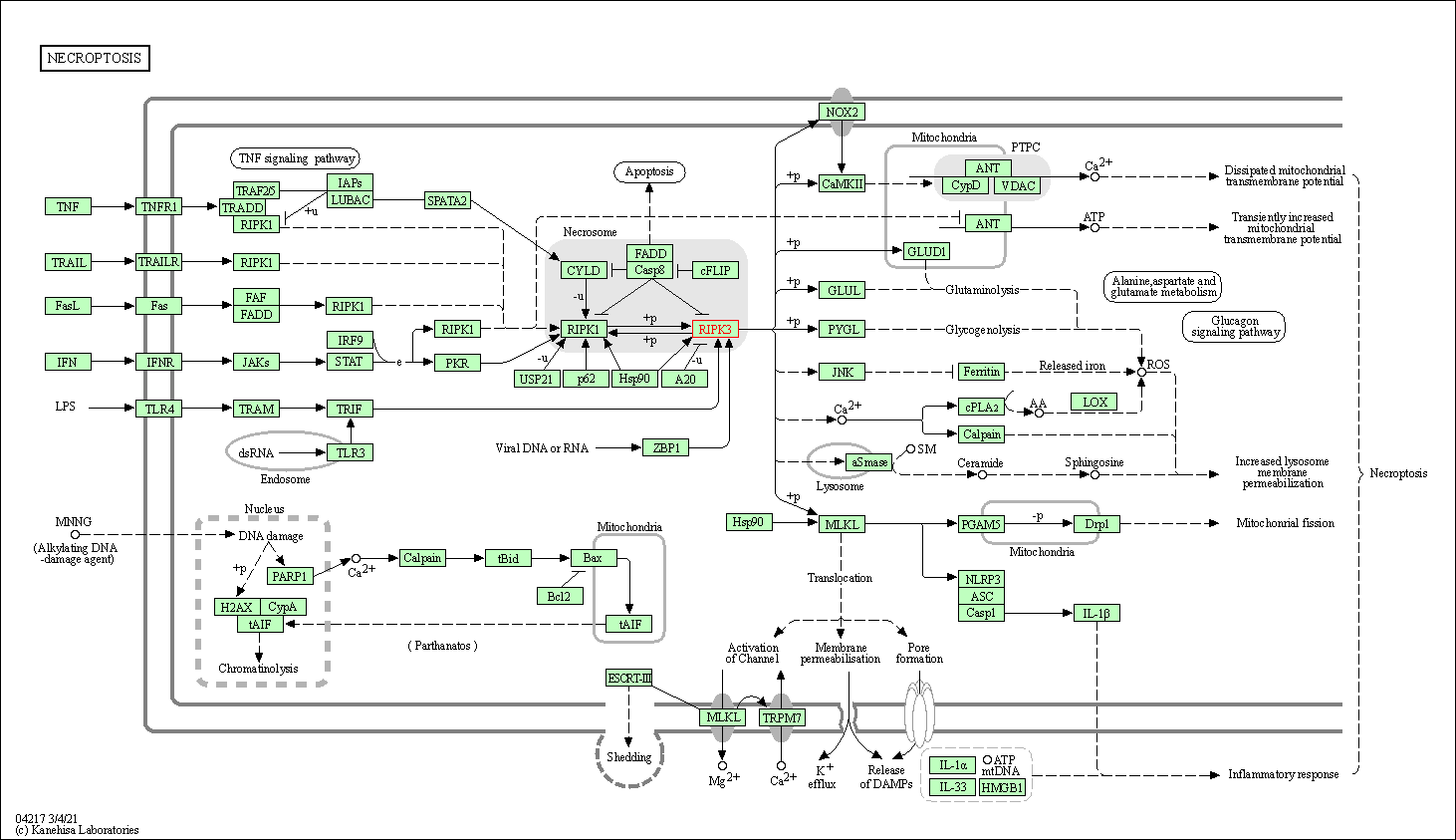
| Up-regulation | Phosphorylation | RIPK3 | Molecule Info |
Pathway MAP
|
||
| In-vitro Model | OVCAR-3 | CVCL_0465 | Ovarian serous adenocarcinoma | Homo sapiens | ||
| Experimental
Result(s) |
The combination therapy of BBR and DDP markedly enhanced more ovarian cancer cell death by inducing apoptosis and necroptosis, which may improve the anticancer effect of chemotherapy drugs. The apoptosis involved the caspase-dependent pathway, while the necroptosis involved the activation of the RIPK3-MLKL pathway. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Telomerase reverse transcriptase (TERT) | Molecule Info | [3] | |
| NetPath Pathway | IL2 Signaling Pathway | Click to Show/Hide | ||
| Pathway Interaction Database | Validated targets of C-MYC transcriptional activation | Click to Show/Hide | ||
| 2 | Regulation of Telomerase | |||
| 3 | IL2 signaling events mediated by PI3K | |||
| 4 | Regulation of nuclear beta catenin signaling and target gene transcription | |||
| 5 | HIF-1-alpha transcription factor network | |||
| Reactome | Formation of the beta-catenin:TCF transactivating complex | Click to Show/Hide | ||