Natural Product (NP) Details
| General Information of the NP (ID: NP4023) | |||||
|---|---|---|---|---|---|
| Name |
Citicoline
|
||||
| Synonyms |
citicoline; cytidine 5'-diphosphocholine; CDP-choline; citicholine; 987-78-0; Citidoline; Cytidoline; Difosfocin; Cyscholin; Haocolin; Nicholin; Niticolin; Recofnan; Recognan; Somazina; Suncholin; Colite; Ensign; cytidine diphosphate choline; Cytidine diphosphocholine; Citicolinum; Citicolina; Cytidine diphosphorylcholine; Choline cytidine diphosphate; Cytidine choline diphosphate; Cytidine 5'-(choline diphosphate); CDP-colina; Cytidine 5'-(cholinyl pyrophosphate); Choline 5'-cytidine diphosphate; cyticholine; Cytidine 5'-diphosphoric choline; Cytidindiphosphocholin; Cereb; Cytidine 5'-diphosphate choline; Cytidine diphosphate choline ester; citidin difosfato de colina; Emicholin; Nicolin; Meibis; CDP-cholin; CDP-choline (neutral charge); Cytidinediphosphoric choline; UNII-536BQ2JVC7; Cytidine 5'-(trihydrogen diphosphate), P'-(2-(trimethylammonio)ethyl) ester, inner salt; IP 302; IP-302; Choline cytidine 5'-pyrophosphate ester; Choline, ester with cytidine 5'-pyrophosphate; CDP-cholin [German]; [2-CYTIDYLATE-O'-PHOSPHONYLOXYL]-ETHYL-TRIMETHYL-AMMONIUM; CDP-colina [Spanish]; Corenalin; Startonyl; Ubelin; Citicoline [INN:JAN]; CYTIDINE-5'-DIPHOSPHOCHOLINE; Citicolinum [INN-Latin]; Citicolina [INN-Spanish]; 536BQ2JVC7; CHEBI:16436; 2-(((((((2R,3S,4R,5R)-5-(4-Amino-2-oxopyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(hydroxy)phosphoryl)oxy)oxidophosphoryl)oxy)-N,N,N-trimethylethanaminium; Cytidindiphosphocholin [German]; Cytidine diphosphate cholin ester; C14H26N4O11P2; EINECS 213-580-7; Citidin difosfato de colina [Spanish]; Cytidine 5-diphosphocholine; NSC 122002; BRN 4170138; Citifar; Hornbest; Neucolis; Sintoclar; Somazine; Reagin; Rexort; Audes; Emicholine F; Cytidine, 5'-pyrophosphate, ester with choline; Cytidine 5'-(trihydrogen diphosphate), P'-[2-(trimethylammonio)ethyl] ester, inner salt; Nicholin (TN); Citicoline (JP17/INN); cytidine-5' diphosphocholine; SCHEMBL221081; AOB5033; Cytidine 5-diphosphate-trihydrogen; MFCD00868097; AKOS025312525; CCG-269597; DB04290; DB12153; Choline, hydroxide, 5'-ester with cytidine 5'-(trihydrogen pyrophosphate), inner salt; cytidine-5'-pyrophosphate-hydroxycholine; [[(2R,3S,4R,5R)-5-(4-amino-2-oxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] 2-(trimethylazaniumyl)ethyl phosphate; Cytidine 5'-(trihydrogen diphosphate), mono(2-(trimethylammonio)ethyl) ester, hydroxide, inner salt; K771; P-hydroxide[2-(trimethylammonio)ethyl] ester; C00307; D00057; W-100063; Q28529682; 5'-O-[(S)-hydroxy({[2-(trimethylammonio)ethoxy]phosphinato}oxy)phosphoryl]cytidine; 5'-O-[hydroxy({[2-(trimethylammonio)ethoxy]phosphinato}oxy)phosphoryl]cytidine; {2-[({[(2R,3S,4R,5R)-5-(4-amino-2-oxo-1,2-dihydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl phosphonato)oxy]ethyl}trimethylazanium
Click to Show/Hide
|
||||
| Species Origin | Escherichia coli ... | Click to Show/Hide | |||
| Escherichia coli | |||||
| Disease | Cerebral ischaemia [ICD-11: 8B1Z] | Approved | [1] | ||
| Structure |
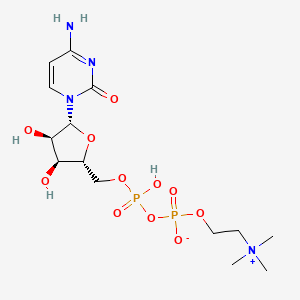
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-5.989
MDCK Permeability
-5.108
PAMPA
+++
HIA
- - -
Distribution
VDss
-0.55
PPB
16.6%
BBB
- - -
Metabolism
CYP1A2 inhibitor
- - -
CYP1A2 substrate
- - -
CYP2C19 inhibitor
- - -
CYP2C19 substrate
- - -
CYP2C9 inhibitor
- - -
CYP2C9 substrate
+
CYP2D6 inhibitor
- - -
CYP2D6 substrate
- - -
CYP3A4 inhibitor
- - -
CYP3A4 substrate
- - -
CYP2B6 inhibitor
- - -
CYP2B6 substrate
- - -
CYP2C8 inhibitor
- - -
HLM Stability
- - -
Excretion
CLplasma
1.716
T1/2
1.769
Toxicity
DILI
+++
Rat Oral Acute Toxicity
+
FDAMDD
+++
Respiratory
+++
Human Hepatotoxicity
- -
Ototoxicity
- - -
Drug-induced Nephrotoxicity
+++
Drug-induced Neurotoxicity
++
Hematotoxicity
- - -
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C14H26N4O11P2
|
||||
| PubChem CID | |||||
| Canonical SMILES |
C[N+](C)(C)CCOP(=O)([O-])OP(=O)(O)OCC1C(C(C(O1)N2C=CC(=NC2=O)N)O)O
|
||||
| InChI |
1S/C14H26N4O11P2/c1-18(2,3)6-7-26-30(22,23)29-31(24,25)27-8-9-11(19)12(20)13(28-9)17-5-4-10(15)16-14(17)21/h4-5,9,11-13,19-20H,6-8H2,1-3H3,(H3-,15,16,21,22,23,24,25)/t9-,11-,12-,13-/m1/s1
|
||||
| InChIKey |
RZZPDXZPRHQOCG-OJAKKHQRSA-N
|
||||
| CAS Number |
CAS 987-78-0
|
||||
| ChEBI ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Hypothermia | Cerebral ischaemia | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vivo Model | Middle cerebral artery occlusion (MCAo) was performed for 2 hours on Sprague-Dawley (SD) rats using intraluminal thread insertion. | |||||
| Experimental
Result(s) |
By suppressing apoptotic processes citicoline with hypothermia is more effective than either used alone in ameliorating cerebral damage after transient focal ischemia. | |||||
| Growth factor | Diminished ovarian reserve | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [3] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Experimental
Result(s) |
Synergistic effects of a low-dose combination of the growth factor bFGF and citicoline after temporary experimental focal cerebral ischemia and furthermore support the effectiveness of a combination treatment regimen for the management of acute stroke. | |||||
| Mexidol | Ischemic stroke | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Experimental
Result(s) |
In patients treated with neipilept or with the combination of citicoline with 2-ethyl-6-methyl-3-hydroxypyridine succinate, plasma phosphatidylcholine was significantly increased. | |||||
| Physiotherapy | Oral cavity carcinoma | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [5] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vivo Model | Clinical Trial | |||||
| Experimental
Result(s) |
Citicoline in combination to physiotherapy appears to be a promising agent to improve gross motor function in patients with cerebral palsy versus physiotherapy alone. | |||||
| Benfotiamine | Substance abuse | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [6] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Experimental
Result(s) |
Co-administration of benfotiamine and citicoline was more effective than either alone in improving memory. | |||||

