Natural Product (NP) Details
| General Information of the NP (ID: NP4040) | |||||
|---|---|---|---|---|---|
| Name |
Chitosan
|
||||
| Synonyms |
Chitosan; Deacetylchitin; Poliglusam; Chicol; Flonac C; Flonac N; Sea Cure Plus; 9012-76-4; Kytex H; Kytex M; Kimitsu Chitosan F; Kimitsu Chitosan H; Kimitsu Chitosan L; Kimitsu Chitosan M; Sea Cure F; Chitopearl 3510; Kimitsu Chitosan F 2; Chitopearl BC 3000; Chitopearl BCW 2500; Chitopearl BCW 3000; Chitopearl BCW 3500; Chitopearl BCW 3505; Chitopearl BCW 3507; CTA 4; YEA; Celox; Poliglusam [USAN:INN]; Dac 70; CCRIS 9144; Chitosan, molecular weight: 100,000-300,000; Chitosan, molecular weight: 600,000-800,000; UNII-82LKS4QV2Y; UNII-SBD1A2I75N; EPA Pesticide Chemical Code 128930; UNII-5GV09YMO52; UNII-7SRJ3W89J8; UNII-23R93M6Y64; Poly (D-glucosamine); 82LKS4QV2Y; SBD1A2I75N; SCHEMBL972215; 5GV09YMO52; 7SRJ3W89J8; SCHEMBL21304990; beta-1,4-Poly-delta-glucosamine; AKOS015918343; 23R93M6Y64; FS-4075; A845562; Q408510; methyl N-[(2S,3R,4R,5S,6R)-5-[(2S,3R,4R,5S,6R)-3-azanyl-5-[(2S,3R,4R,5S,6R)-3-azanyl-5-[(2S,3R,4R,5S,6R)-3-azanyl-5-[(2S,3R,4R,5S,6R)-3-azanyl-5-[(2S,3R,4R,5S,6R)-3-azanyl-5-[(2S,3R,4R,5S,6R)-3-azanyl-6-(hydroxymethyl)-4,5-bis(oxidanyl)oxan-2-yl]oxy-6-(hy; N-[(2S,3R,4R,5S,6R)-5-[[(2S,3R,4R,5S,6R)-3-amino-5-[[(2S,3R,4R,5S,6R)-3-amino-5-[[(2S,3R,4R,5S,6R)-3-amino-5-[[(2S,3R,4R,5S,6R)-3-amino-5-[[(2S,3R,4R,5S,6R)-3-amino-5-[[(2S,3R,4R,5S,6R)-3-amino-4,5-dihydroxy-6-(hydroxymethyl)-2-oxanyl]oxy]-4-hydroxy-6-(hy
Click to Show/Hide
|
||||
| Species Origin | Lepidurus arcticus ... | Click to Show/Hide | |||
| Lepidurus arcticus | |||||
| Disease | Prostate cancer [ICD-11: 2C82] | Phase 2 | [1] | ||
| Structure |
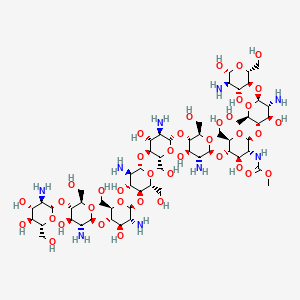
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-7.949
MDCK Permeability
-4.812
PAMPA
+++
HIA
+++
Distribution
VDss
-0.541
PPB
5.4%
BBB
- - -
Metabolism
CYP1A2 inhibitor
- - -
CYP1A2 substrate
- - -
CYP2C19 inhibitor
- - -
CYP2C19 substrate
+++
CYP2C9 inhibitor
- - -
CYP2C9 substrate
- - -
CYP2D6 inhibitor
- - -
CYP2D6 substrate
- - -
CYP3A4 inhibitor
- - -
CYP3A4 substrate
- - -
CYP2B6 inhibitor
- - -
CYP2B6 substrate
- - -
CYP2C8 inhibitor
- - -
HLM Stability
- - -
Excretion
CLplasma
-1.763
T1/2
4.728
Toxicity
DILI
+++
Rat Oral Acute Toxicity
- - -
FDAMDD
- - -
Respiratory
- - -
Human Hepatotoxicity
+++
Ototoxicity
+++
Drug-induced Nephrotoxicity
+++
Drug-induced Neurotoxicity
- - -
Hematotoxicity
+++
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C56H103N9O39
|
||||
| PubChem CID | |||||
| Canonical SMILES |
COC(=O)NC1C(C(C(OC1OC2C(OC(C(C2O)N)OC3C(OC(C(C3O)N)O)CO)CO)CO)OC4C(C(C(C(O4)CO)OC5C(C(C(C(O5)CO)OC6C(C(C(C(O6)CO)OC7C(C(C(C(O7)CO)OC8C(C(C(C(O8)CO)OC9C(C(C(C(O9)CO)O)O)N)O)N)O)N)O)N)O)N)O)N)O
|
||||
| InChI |
1S/C56H103N9O39/c1-87-56(86)65-28-38(84)46(19(10-74)96-55(28)104-45-18(9-73)95-49(27(64)37(45)83)97-39-12(3-67)88-47(85)20(57)31(39)77)103-54-26(63)36(82)44(17(8-72)94-54)102-53-25(62)35(81)43(16(7-71)93-53)101-52-24(61)34(80)42(15(6-70)92-52)100-51-23(60)33(79)41(14(5-69)91-51)99-50-22(59)32(78)40(13(4-68)90-50)98-48-21(58)30(76)29(75)11(2-66)89-48/h11-55,66-85H,2-10,57-64H2,1H3,(H,65,86)/t11-,12-,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23-,24-,25-,26-,27-,28-,29-,30-,31-,32-,33-,34-,35-,36-,37-,38-,39-,40-,41-,42-,43-,44-,45-,46-,47-,48+,49+,50+,51+,52+,53+,54+,55+/m1/s1
|
||||
| InChIKey |
FLASNYPZGWUPSU-SICDJOISSA-N
|
||||
| CAS Number |
CAS 9012-76-4
|
||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Erythromycin | Bacterial infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Propionibacterium acnes KCTC 3314 | Microorganism model | Propionibacterium acnes | |||
| Staphylococcus aureus KCTC 1927 | Microorganism model | Staphylococcus aureus | ||||
| Staphylococcus epidermidis KCTC 1370 | Microorganism model | Staphylococcus epidermidis | ||||
| Pseudomonas aeruginosa KCTC 1637 | Microorganism model | Pseudomonas aeruginosa | ||||
| Experimental
Result(s) |
Among chitosan-conjugated derivatives, Chitosan-caffeic acid showed the highest antibacterial activity and also exhibited the synergistic antibacterial effect in combination with tetracycline, erythromycin, and lincomycin against acne-related bacteria. | |||||
| Tetracycline | Bacterial infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Propionibacterium acnes KCTC 3314 | Microorganism model | Propionibacterium acnes | |||
| Staphylococcus aureus KCTC 1927 | Microorganism model | Staphylococcus aureus | ||||
| Staphylococcus epidermidis KCTC 1370 | Microorganism model | Staphylococcus epidermidis | ||||
| Pseudomonas aeruginosa KCTC 1637 | Microorganism model | Pseudomonas aeruginosa | ||||
| Experimental
Result(s) |
Among chitosan-conjugated derivatives, Chitosan-caffeic acid showed the highest antibacterial activity and also exhibited the synergistic antibacterial effect in combination with tetracycline, erythromycin, and lincomycin against acne-related bacteria. | |||||
| Lincomycin | Gram-positive bacterial infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Propionibacterium acnes KCTC 3314 | Microorganism model | Propionibacterium acnes | |||
| Staphylococcus aureus KCTC 1927 | Microorganism model | Staphylococcus aureus | ||||
| Staphylococcus epidermidis KCTC 1370 | Microorganism model | Staphylococcus epidermidis | ||||
| Pseudomonas aeruginosa KCTC 1637 | Microorganism model | Pseudomonas aeruginosa | ||||
| Experimental
Result(s) |
Among chitosan-conjugated derivatives, Chitosan-caffeic acid showed the highest antibacterial activity and also exhibited the synergistic antibacterial effect in combination with tetracycline, erythromycin, and lincomycin against acne-related bacteria. | |||||

