Natural Product (NP) Details
| General Information of the NP (ID: NP4551) | |||||
|---|---|---|---|---|---|
| Name |
Heparin
|
||||
| Synonyms |
heparin; enoxaparin; 9005-49-6; Ardeparin; Bemiparin; SEMULOPARIN; Lovenox; LMWH; Nadroparin; Nadroparine; Clivarin; Liquaemin; Reviparin; 6-((5-acetamido-4,6-dihydroxy-2-((sulfooxy)methyl)tetrahydro-2H-pyran-3-yl)oxy)-3-((5-((6-carboxy-4,5-dihydroxy-3-(sulfooxy)tetrahydro-2H-pyran-2-yl)oxy)-6-(hydroxymethyl)-3-(sulfoamino)-4-(sulfooxy)tetrahydro-2H-pyran-2-yl)oxy)-4-hydroxy-5-(sulfooxy)tetrahydro-2H-pyran-2-carboxylic acid; Adomiparin; Multiparin; Novoheparin; Parnaparin; Parvoparin; Sandoparin; Sublingula; Tinzaparin; Heparina; Heparine; Heparinum; Hepathrom; Liquemin; Octaparin; Pabyrin; PK-10169; Pularin; Subeparin; Triofiban; Fluxum; Vetren; Hed-heparin; Depo-Heparin; Lipo-hepin; Fragmin A; Fragmin B; Vitrum AB; Eparina [DCIT]; Heparin [BAN]; Adomiparin [USAN]; CY 216; FR 860; Heparin CY 216; Heparine [INN-French]; Heparinum [INN-Latin]; Heparina [INN-Spanish]; Semuloparin [USAN:INN]; UNII-1K5KDI46KZ; UNII-4QW4AN84NQ; UNII-E47C0NF7LV; UNII-VL0L558GCB; 1K5KDI46KZ; 4QW4AN84NQ; E47C0NF7LV; UNII-P776JQ4R2F; UNII-V72OT3K19I; VL0L558GCB; 2-o-sulfohexopyranuronosyl-(1->4)-2-deoxy-3-o-sulfo-2-(sulfoamino)hexopyranosyl-(1->4)-2-o-sulfohexopyranuronosyl-(1->4)-2-acetamido-2-deoxy-6-o-sulfohexopyranose; SCHEMBL543122; UNII-5R0L1D739E; UNII-7UQ7X4Y489; UNII-M316WT19D8; UNII-S79O08V79F; UNII-T2410KM04A; GTPL6811; M 118REH; P776JQ4R2F; V72OT3K19I; Cy 222; HSDB 3094; DTXSID80872762; 5R0L1D739E; 7UQ7X4Y489; M316WT19D8; S79O08V79F; T2410KM04A; UNII-9816XA9004; AVE-5026; BCP13334; EINECS 232-681-7; KB 101; OP 386; OP 622; RP-54563; 9816XA9004; M 118; Q416516; 6-[5-acetamido-4,6-dihydroxy-2-(sulfooxymethyl)oxan-3-yl]oxy-3-[5-(6-carboxy-4,5-dihydroxy-3-sulfooxyoxan-2-yl)oxy-6-(hydroxymethyl)-3-(sulfoamino)-4-sulfooxyoxan-2-yl]oxy-4-hydroxy-5-sulfooxyoxane-2-carboxylic acid; 6-[6-[6-[5-acetamido-4,6-dihydroxy-2-(sulfooxymethyl)tetrahydropyran-3-yl]oxy-2-carboxy-4-hydroxy-5-sulfooxy-tetrahydropyran-3-yl]oxy-2-(hydroxymethyl)-5-(sulfoamino)-4-sulfooxy-tetrahydropyran-3-yl]oxy-3,4-dihydroxy-5-sulfooxy-tetrahydropyran-2-carboxylic acid
Click to Show/Hide
|
||||
| Species Origin | Filipendula ulmaria ... | Click to Show/Hide | |||
| Filipendula ulmaria | |||||
| Disease | Deep vein thrombosis [ICD-11: BD71] | Approved | [1] | ||
| Structure |
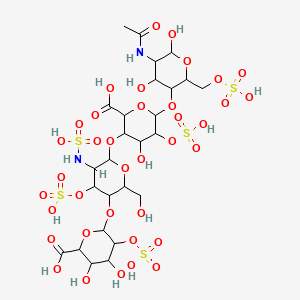
|
Click to Download Mol2D MOL |
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C26H42N2O37S5
|
||||
| PubChem CID | |||||
| Canonical SMILES |
CC(=O)NC1C(C(C(OC1O)COS(=O)(=O)O)OC2C(C(C(C(O2)C(=O)O)OC3C(C(C(C(O3)CO)OC4C(C(C(C(O4)C(=O)O)O)O)OS(=O)(=O)O)OS(=O)(=O)O)NS(=O)(=O)O)O)OS(=O)(=O)O)O
|
||||
| InChI |
1S/C26H42N2O37S5/c1-4(30)27-7-9(31)13(6(56-23(7)39)3-55-67(43,44)45)58-26-19(65-70(52,53)54)12(34)16(20(62-26)22(37)38)60-24-8(28-66(40,41)42)15(63-68(46,47)48)14(5(2-29)57-24)59-25-18(64-69(49,50)51)11(33)10(32)17(61-25)21(35)36/h5-20,23-26,28-29,31-34,39H,2-3H2,1H3,(H,27,30)(H,35,36)(H,37,38)(H,40,41,42)(H,43,44,45)(H,46,47,48)(H,49,50,51)(H,52,53,54)
|
||||
| InChIKey |
HTTJABKRGRZYRN-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 9005-49-6
|
||||
| Herb ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| X-ray irradiation | Scaphoid Fracture | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Down-regulation | Expression | BIRC5 | Molecule Info |
Pathway MAP
|
|
| Down-regulation | Expression | CD34 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | IL6 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | TGFB1 | Molecule Info |
Pathway MAP
|
||
| In-vivo Model | Lewis lung cancer cells (~3*106) were subcutaneously injected into the right anterior leg of each mouse to establish the tumor-bearing mouse model. | |||||
| Experimental
Result(s) |
A combinatory treatment strategy of nadroparin with fractionated irradiation had a strong synergistic antitumor effect in vivo, which may be associated with the promotion of apoptosis, inhibited secretion of TGF-Beta1 and IL-6 and down-regulation of CD34 and survivin expression. | |||||
| Bivalirudin | Thrombocytopenia | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [3] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Biological
Regulation |
Decrease | clotting time | ||||
| In-vitro Model | Human fibroblasts collected from a skin biopsy | Healthy | Homo sapiens | |||
| Human liver non-parenchymal cells obtained after liver isolation | Healthy | Homo sapiens | ||||
| hALPCs obtained from healthy liver donors | Healthy | Homo sapiens | ||||
| Experimental
Result(s) |
Concomitant administration of an antithrombin activator or direct factor Xa inhibitor and direct thrombin inhibitor proved to be a particularly effective combination for controlling the procoagulant effects of hALPCs both in vitro and in vivo. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Antithrombin-III (ATIII) | Molecule Info | [4] | |
| KEGG Pathway | Complement and coagulation cascades | Click to Show/Hide | ||
| Panther Pathway | Blood coagulation | Click to Show/Hide | ||
| Pathway Interaction Database | Glypican 1 network | Click to Show/Hide | ||
| Reactome | Intrinsic Pathway of Fibrin Clot Formation | Click to Show/Hide | ||
| 2 | Common Pathway of Fibrin Clot Formation | |||
| WikiPathways | Complement and Coagulation Cascades | Click to Show/Hide | ||
| 2 | Formation of Fibrin Clot (Clotting Cascade) | |||