Natural Product (NP) Details
| General Information of the NP (ID: NP6495) | |||||
|---|---|---|---|---|---|
| Name |
Fluoroorotic acid
|
||||
| Synonyms |
5-Fluoroorotic acid; 703-95-7; 5-fluoro-2,6-dioxo-1,2,3,6-tetrahydropyrimidine-4-carboxylic acid; 5-Fluoroorotate; Fluoroorotic acid; Orotic acid, 5-fluoro-; 5-Fluorouracil-4-carboxylic acid; 5-FOA; ENT-26398; NSC 31712; Ro 2-9945; UNII-7IA9OUC93E; WR 152520; MLS000737636; 5-fluoro-2,4-dioxo-1H-pyrimidine-6-carboxylic acid; 4-Pyrimidinecarboxylic acid, 5-fluoro-1,2,3,6-tetrahydro-2,6-dioxo-; 7IA9OUC93E; Fluoroorotic Acid, Ultra Pure; FOA; 5-Fluorouracil-6-carboxylic Acid; 1,2,3,6-Tetrahydro-2,6-dioxo-5-fluoro-4-pyrimidinecarboxylic acid; MFCD00042526; SMR000393806; 4-Pyrimidinecarboxylic acid, 1,2,3,6-tetrahydro-2,6-dioxo-5-fluoro-; 5-Fluoro orotic acid; 5-Fluoroorotic acid (VAN); EINECS 211-876-0; 5-fluoro-2,6-dioxo-1,3-dihydropyrimidine-4-carboxylic acid; AI3-26398; 5-Fluoroortic acid; FOT; 174.09 (anhydrous); C5H3FN2O4.xH2O; 5-Fluoroorotic acid;5-FOA; SCHEMBL44527; WLN: T6VMVMJ EVQ FF; cid_69711; CHEMBL1232805; BDBM47470; 5-Fluoroorotic acid, 98.0%+; DTXSID90220573; 5-fluorouracil-4-carboxylic acid;; HMS2759G19; ACT03077; EBD39533; NSC31712; ZINC1663959; 5-Fluoro-1,2,3,6-tetrahydro-2,6-dioxo-4-pyrimidinecarboxylic acid; ANW-46244; NSC-31712; SBB066881; AKOS005207200; CS-W017535; 4-Pyrimidinecarboxylic acid, 5-fluoro-1,2,3,6-tetrahydro-2,6-dioxo-, monohydrate; 5-Fluorouracil-4-carboxylic acid hydrate; NCGC00246927-01; Orotic acid, 5-fluoro- (VAN) (8CI); AC-10068; AS-14168; K187; SY001827; AB0014081; F0382; FT-0620427; ST50405211; F-6300; M-2738; 45988-EP2298735A1; 45988-EP2311807A1; 703F957; 2,6-Dihydroxy-5-fluoropyrimidine-4-carboxylic acid; 5-fluoro-2,4-diketo-1H-pyrimidine-6-carboxylic acid; Q18207160; 1,3,6-Tetrahydro-2,6-dioxo-5-fluoro-4-pyrimidinecarboxylic acid; 4-Pyrimidinecarboxylic acid,2,3,6-tetrahydro-2,6-dioxo-5-fluoro-; 5-fluoranyl-2,4-bis(oxidanylidene)-1H-pyrimidine-6-carboxylic acid; 5-Fluoro-2,6-dioxo-1,2,3,6-tetrahydro-4-pyrimidinecarboxylic acid; 5-Fluoro-2,6-dioxo-1,2,3,6-tetrahydro-pyrimidine-4-carboxylic acid; 4-Pyrimidinecarboxylic acid, 5-fluoro-1,2,3,6-tetrahydro-2,6-dioxo- (9CI)
Click to Show/Hide
|
||||
| Species Origin | Homo sapiens ... | Click to Show/Hide | |||
| Homo sapiens | |||||
| Disease | Aspergillosis [ICD-11: 1F20] | Investigative | [1] | ||
| Structure |
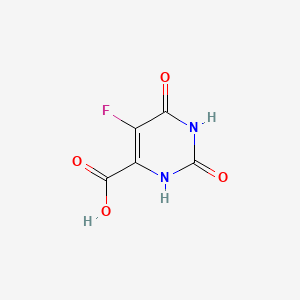
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-5.644
MDCK Permeability
-4.767
PAMPA
+++
HIA
++
Distribution
VDss
-0.538
PPB
50.1%
BBB
- - -
Metabolism
CYP1A2 inhibitor
- - -
CYP1A2 substrate
- - -
CYP2C19 inhibitor
- - -
CYP2C19 substrate
- - -
CYP2C9 inhibitor
- - -
CYP2C9 substrate
- - -
CYP2D6 inhibitor
- - -
CYP2D6 substrate
- - -
CYP3A4 inhibitor
- - -
CYP3A4 substrate
- - -
CYP2B6 inhibitor
- - -
CYP2B6 substrate
- - -
CYP2C8 inhibitor
- - -
HLM Stability
- - -
Excretion
CLplasma
2.155
T1/2
2.175
Toxicity
DILI
+++
Rat Oral Acute Toxicity
+
FDAMDD
- - -
Respiratory
++
Human Hepatotoxicity
+++
Ototoxicity
+++
Drug-induced Nephrotoxicity
+++
Drug-induced Neurotoxicity
+++
Hematotoxicity
+
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C5H3FN2O4
|
||||
| PubChem CID | |||||
| Canonical SMILES |
C1(=C(NC(=O)NC1=O)C(=O)O)F
|
||||
| InChI |
1S/C5H3FN2O4/c6-1-2(4(10)11)7-5(12)8-3(1)9/h(H,10,11)(H2,7,8,9,12)
|
||||
| InChIKey |
SEHFUALWMUWDKS-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 703-95-7
|
||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Uftoral + Oxaliplatin | Click to Show/Hide the Molecular Data of This Drug | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | HT-29 | CVCL_0320 | Colon adenocarcinoma | Homo sapiens | ||
| In-vivo Model | Human colorectal HT29 cell xenografts (1*106 cells) were transplanted s.c. into the hind limb of each animal (Female BALB/c nude mice (nu+/nu+) aged 6 weeks). | |||||
| Experimental
Result(s) |
Combined treatment (UFT/FA+oxaliplatin) reduced tumor weight by 39% compared to oxaliplatin alone (p<0.05) or UFT/FA (p<0.05). | |||||
| Oxaliplatin + 5-fluorouracil | Click to Show/Hide the Molecular Data of This Drug | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [3] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vivo Model | Clinical trial | |||||
| Experimental
Result(s) |
Combination chemotherapy with oxaliplatin, 5-FU, and FA is an active and well-tolerated regimen as first-line treatment in patients with metastatic or recurrent gastric cancer. | |||||
| Cisplatin + 5-fluorouracil | Click to Show/Hide the Molecular Data of This Drug | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Experimental
Result(s) |
Weekly administration of 5-FU/FA/cisplatin is an active and well-tolerated regimen. Toxicity is manageable and allows chemotherapy on an outpatient basis without hydration program as required when cisplatin is used at the dose of 50 mg/m2. | |||||
| Raltitrexed + 5-fluorouracil | Click to Show/Hide the Molecular Data of This Drug | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [5] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | LoVo | CVCL_0399 | Colon adenocarcinoma | Homo sapiens | ||
| HT-29 | CVCL_0320 | Colon adenocarcinoma | Homo sapiens | |||
| Experimental
Result(s) |
Histone deacetylase inhibitor NVP-LAQ824 sensitizes human nonsmall cell lung cancer to the cytotoxic effects of ionizing radiation. | |||||
| Methioninase + 5-fluorouracil | Click to Show/Hide the Molecular Data of This Drug | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [6] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | CCRF-CEM | CVCL_0207 | T acute lymphoblastic leukemia | Homo sapiens | ||
| Experimental
Result(s) |
Methioninase in combination with 5-fluorouracil and folinic acid showed cytotoxic synergism in CCRF-CEM. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Plasmodium dihydroorotase (Malaria dho) | Molecule Info | [7] | |
| Plasmodium dihydroorotate dehydrogenase (Malaria DHOdehase) | Molecule Info | [7] | ||
| KEGG Pathway | Pyrimidine metabolism | Click to Show/Hide | ||
| 2 | Metabolic pathways | |||
| Pathwhiz Pathway | Pyrimidine Metabolism | Click to Show/Hide | ||
| Reactome | Pyrimidine biosynthesis | Click to Show/Hide | ||
| WikiPathways | Metabolism of nucleotides | Click to Show/Hide | ||

