Natural Product (NP) Details
| General Information of the NP (ID: NP9436) | |||||
|---|---|---|---|---|---|
| Name |
Camptothecin
|
||||
| Synonyms |
camptothecin; Camptothecine; (S)-(+)-Camptothecin; 7689-03-4; Campathecin; (+)-Camptothecine; d-Camptothecin; (+)-Camptothecin; 20(S)-Camptothecine; 21,22-Secocamptothecin-21-oic acid lactone; NSC94600; Camptothecine (8CI); Camptothecine (S,+); CHEMBL65; (4S)-4-ethyl-4-hydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; (S)-4-ethyl-4-hydroxy-1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; UNII-XT3Z54Z28A; MLS000766223; XT3Z54Z28A; CHEBI:27656; MFCD00081076; NSC-94600; (+)-Camptothecin, 98%; (S)-Camptothecin; 1H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione, 4-ethyl-4-hydroxy-, (S)-; 20(S)-Camptothecin; 4-ETHYL-4-HYDROXY-1,12-DIHYDRO-4H-2-OXA-6,12A-DIAZA-DIBENZO[B,H]FLUORENE-3,13-DIONE; SR-01000075798; SR-01000597379; d-camptothecine; (s)-camptothecine; Camptothecin, Camptotheca acuminata; Camptothecin,(S); ( )-Camptothecin; (S)-4-ethyl-4-hydroxy-1H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione; (S)-4-Ethyl-4-hydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14-(4H,12H)-dione; 1H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione, 4-ethyl-4-hydroxy-, (4S)-; 1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione, 4-ethyl-4-hydroxy-, (4S)-; 1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione, 4-ethyl-4-hydroxy-, (S)-; Prestwick_102; (+)-Camptothecin;; PubChem10183; Spectrum_000299; Tocris-1100; SpecPlus_000712; Prestwick0_000200; Prestwick1_000200; Prestwick2_000200; Prestwick3_000200; Spectrum2_000903; Spectrum3_001203; Spectrum4_000738; Spectrum5_001126; Lopac-C-9911; SCHEMBL6038; Lopac0_000341; BSPBio_000159; BSPBio_002586; KBioGR_001036; KBioSS_000779; KBioSS_002283; cid_24360; DivK1c_000826; DivK1c_006808; SPECTRUM1502232; SPBio_000746; SPBio_002080; BPBio1_000175; CCRIS 8162; DTXSID0030956; HMS502J08; KBio1_000826; KBio1_001752; KBio2_000779; KBio2_003347; KBio2_005915; KBio3_002086; 4-Ethyl-4-hydroxy-1H-pyrano-[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; NINDS_000826; Bio1_000400; Bio1_000889; Bio1_001378; HMS1568H21; HMS1921N08; HMS2089F08; HMS2095H21; HMS3261E03; HMS3414J17; HMS3654D13; HMS3678J15; HMS3712H21; ZINC105309; (S)-4-Ethyl-4-hydroxy-1H-pyrano-[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; ACT02668; BCP02857; Tox21_500341; AC-202; BBL033963; BDBM50008923; CCG-40255; GR-301; NSC 94600; s1288; STK801886; AKOS004119861; CS-1049; DB04690; KS-5235; LP00341; MCULE-1654983682; SDCCGMLS-0066688.P001; SDCCGSBI-0050329.P003; BRN 0631069; CAS-2114454; IDI1_000826; NCGC00015290-01; NCGC00016994-01; NCGC00016994-02; NCGC00016994-03; NCGC00016994-04; NCGC00016994-05; NCGC00016994-06; NCGC00016994-07; NCGC00016994-08; NCGC00016994-09; NCGC00016994-10; NCGC00016994-11; NCGC00016994-12; NCGC00016994-16; NCGC00016994-29; NCGC00024997-01; NCGC00024997-02; NCGC00024997-03; NCGC00024997-04; NCGC00024997-05; NCGC00024997-06; NCGC00178592-01; NCGC00178592-02; NCGC00261026-01; AK-39842; HY-16560; K332; NCI60_042105; SMR000445686; SY010324; AI3-62475; EU-0100341; N1868; SW196414-3; C 9911; C01897; J10012; M01564; S-2515; 32320-EP2270008A1; 32320-EP2272827A1; 32320-EP2275420A1; 32320-EP2277507A1; 32320-EP2277865A1; 32320-EP2280013A1; 32320-EP2281815A1; 32320-EP2286795A1; 32320-EP2286812A1; 32320-EP2289892A1; 32320-EP2292615A1; 32320-EP2292617A1; 32320-EP2295055A2; 32320-EP2295416A2; 32320-EP2298748A2; 32320-EP2298764A1; 32320-EP2298765A1; 32320-EP2298778A1; 32320-EP2301933A1; 32320-EP2305640A2; 32320-EP2305642A2; 32320-EP2305671A1; 32320-EP2308855A1; 32320-EP2311453A1; 32320-EP2311827A1; 32320-EP2311840A1; AB00052452-08; AB00052452-09; AB00052452_10; (S)-(+)-Camptothecin, >=90% (HPLC), powder; 157594-EP2277507A1; 157594-EP2286795A1; 179002-EP2280013A1; 689C034; Q419964; Q-200785; SR-01000075798-1; SR-01000075798-4; SR-01000597379-1; SR-01000597379-3; BRD-K37890730-001-09-4; BRD-K37890730-001-10-2; (S)-4-ethyl-4-hydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]florene-3,13-dione; (S)-4-ethyl-4-hydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione; 4-Ethyl-4-hydroxy-1H-pyrano-[3[,4[:6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; Topotecan Related Compound C, United States Pharmacopeia (USP) Reference Standard; (19S)-19-ethyl-19-hydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.0(2),(1)(1).0?,?.0(1)?,(2)?]henicosa-1(21),2,4,6,8,10,15(20)-heptaene-14,18-dione; (S)-4-Ethyl-4-hydroxy-1H-pyrano[3 inverted exclamation mark ,4 inverted exclamation mark :6,7]indolizino[1,2-b]quinoline-3,14-(4H,12H)-dione; 1H-Pyrano[3',7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione, 4-ethyl-4-hydroxy-, (S)-; 4(S)-Ethyl-4-hydroxy-1H-pyrano-[3',4':6,7]indolizino[1,2-b]quinoline-3,14 (4H,12H)-dione; 4-ethyl-4-hydroxy-(4S)-3,4,12,14-tetrahydro-1H-pyrano[3'',4'':6,7]indolizino[1,2-b]quinoline-3,14-dione; 4-Ethyl-4-hydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione (camptothecin or CPT); 4-Ethyl-4-hydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione (Camptothecin); 4-Ethyl-4-hydroxy-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione (CPT, Camptothecin)
Click to Show/Hide
|
||||
| Species Origin | Camptotheca acuminata ... | Click to Show/Hide | |||
| Camptotheca acuminata | |||||
| Disease | Solid tumour/cancer [ICD-11: 2A00-2F9Z] | Phase 3 | [1] | ||
| Structure |
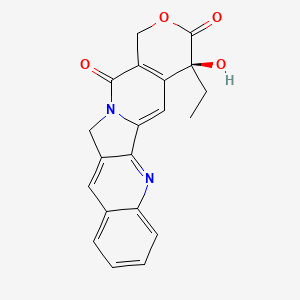
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-5.091
MDCK Permeability
-4.676
PAMPA
+++
HIA
- - -
Distribution
VDss
-0.366
PPB
98.9%
BBB
-
Metabolism
CYP1A2 inhibitor
+
CYP1A2 substrate
++
CYP2C19 inhibitor
- - -
CYP2C19 substrate
- - -
CYP2C9 inhibitor
- - -
CYP2C9 substrate
- - -
CYP2D6 inhibitor
- - -
CYP2D6 substrate
- - -
CYP3A4 inhibitor
- -
CYP3A4 substrate
++
CYP2B6 inhibitor
- - -
CYP2B6 substrate
- - -
CYP2C8 inhibitor
++
HLM Stability
+++
Excretion
CLplasma
6.248
T1/2
1.497
Toxicity
DILI
++
Rat Oral Acute Toxicity
++
FDAMDD
+++
Respiratory
- -
Human Hepatotoxicity
+++
Ototoxicity
+
Drug-induced Nephrotoxicity
+++
Drug-induced Neurotoxicity
+++
Hematotoxicity
+++
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C20H16N2O4
|
||||
| PubChem CID | |||||
| Canonical SMILES |
CCC1(C2=C(COC1=O)C(=O)N3CC4=CC5=CC=CC=C5N=C4C3=C2)O
|
||||
| InChI |
1S/C20H16N2O4/c1-2-20(25)14-8-16-17-12(7-11-5-3-4-6-15(11)21-17)9-22(16)18(23)13(14)10-26-19(20)24/h3-8,25H,2,9-10H2,1H3/t20-/m0/s1
|
||||
| InChIKey |
VSJKWCGYPAHWDS-FQEVSTJZSA-N
|
||||
| CAS Number |
CAS 7689-03-4
|
||||
| ChEBI ID | |||||
| Herb ID | |||||
| SymMap ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Doxorubicin | Solid tumour/cancer | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Up-regulation | Expression | CTNNB1 | Molecule Info |
Pathway MAP
|
|
| In-vitro Model | 4T1 | CVCL_0125 | Malignant neoplasms | Mus musculus | ||
| BT-474 | CVCL_0179 | Invasive breast carcinoma | Homo sapiens | |||
| In-vivo Model | Tumor inoculation was achieved by subcutaneously injecting 5*104 4T1 cells in the abdominal mammary gland of female BALB/c mice in order to prevent interference with normal bodily functions | |||||
| Experimental
Result(s) |
Camptothecin-doxorubicin combinations showed synergistic antitumor acitivity. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | DNA topoisomerase I (TOP1) | Molecule Info | [1] | |
| NetPath Pathway | IL2 Signaling Pathway | Click to Show/Hide | ||
| Panther Pathway | DNA replication | Click to Show/Hide | ||
| Pathway Interaction Database | Caspase Cascade in Apoptosis | Click to Show/Hide | ||
| WikiPathways | Integrated Pancreatic Cancer Pathway | Click to Show/Hide | ||