Natural Product (NP) Details
| General Information of the NP (ID: NP9882) | |||||
|---|---|---|---|---|---|
| Name |
Anidulafungin
|
||||
| Synonyms |
Anidulafungin; Eraxis; 166663-25-8; Ecalta; UNII-9HLM53094I; Anidulafungin (LY303366); VER-002; CHEBI:55346; LY-303366; 9HLM53094I; LY303366; V-Echinocandin; Anidulafungina; Anidulafungine; Anidulafunginum; Anidulafungin [USAN:INN:BAN]; C58H73N7O17; SCHEMBL38292; (4R,5R)-4,5-Dihydroxy-N(sup 2)-((4'-(pentyloxy)-p-terphenyl-4-yl)carbonyl)-L-ornithyl-L-threonyl-trans-4-hydroxy-L-prolyl-(S)-4-hydroxy-4-(p-hydroxyphenyl)-L-threonyl-L-threonyl-(3S,4S)-3-hydroxy-4-methyl-L-proline cyclic (6-1)-peptide; CHEMBL264241; VER002; BDBM50417554; MFCD00917070; s4286; CCG-270647; DB00362; NCGC00386349-01; AT-36943; SW222238-1; A13436; D70013; Z-3192; 663A258; J-010304; Q4764531; Echinocandin B, 1-((4R,5R)-4,5-dihydroxy-N2-((4''-(pentyloxy)(1,1':4',1''-terphenyl)-4-yl)carbonyl)-L-ornithine); N-[(3S,6S,9S,11R,15S,18S,20R,21R,24S,25S,26S)-6-[(1S,2S)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl]-11,20,21,25-tetrahydroxy-3,15-bis[(1R)-1-hydroxyethyl]-26-methyl-2,5,8,14,17,23-hexaoxo-1,4,7,13,16,22-hexaazatricyclo[22.3.0.0^{9,13}]heptacosan-18-yl]-4-{4-[4-(pentyloxy)phenyl]phenyl}benzamide; N-[[(1S,2S)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl]-tetrahydroxy-bis[(1R)-1-hydroxyethyl]-methyl-hexaoxo-[?]yl]-4-[4-(4-pentoxyphenyl)phenyl]benzamide; N-{(2R,6S,9S,11R,12R,14aS,15S,16S,20S,23S,25aS)-23-[(1S,2S)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl]-2,11,12,15-tetrahydroxy-6,20-bis[(1R)-1-hydroxyethyl]-16-methyl-5,8,14,19,22,25-hexaoxotetracosahydro-1H-dipyrrolo[2,1-c:2',1'-l][1,4,7,10,13,16]hexaazacyclohenicosin-9-yl}-4''-(pentyloxy)-1,1':4',1''-terphenyl-4-carboxamide
Click to Show/Hide
|
||||
| Species Origin | Aspergillus nidulans ... | Click to Show/Hide | |||
| Aspergillus nidulans | |||||
| Disease | Epilepsy/seizure [ICD-11: 8A60] | Approved | [1] | ||
| Structure |
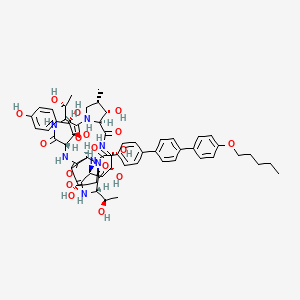
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-6.445
MDCK Permeability
-5.418
PAMPA
+++
HIA
+++
Distribution
VDss
-0.418
PPB
95.2%
BBB
- - -
Metabolism
CYP1A2 inhibitor
- - -
CYP1A2 substrate
- - -
CYP2C19 inhibitor
- - -
CYP2C19 substrate
- - -
CYP2C9 inhibitor
- - -
CYP2C9 substrate
- - -
CYP2D6 inhibitor
- - -
CYP2D6 substrate
- - -
CYP3A4 inhibitor
- - -
CYP3A4 substrate
- - -
CYP2B6 inhibitor
+++
CYP2B6 substrate
- - -
CYP2C8 inhibitor
+++
HLM Stability
++
Excretion
CLplasma
1.132
T1/2
3.388
Toxicity
DILI
+++
Rat Oral Acute Toxicity
- - -
FDAMDD
- -
Respiratory
- - -
Human Hepatotoxicity
+++
Ototoxicity
+++
Drug-induced Nephrotoxicity
+++
Drug-induced Neurotoxicity
- - -
Hematotoxicity
- - -
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C58H73N7O17
|
||||
| PubChem CID | |||||
| Canonical SMILES |
CCCCCOC1=CC=C(C=C1)C2=CC=C(C=C2)C3=CC=C(C=C3)C(=O)NC4CC(C(NC(=O)C5C(C(CN5C(=O)C(NC(=O)C(NC(=O)C6CC(CN6C(=O)C(NC4=O)C(C)O)O)C(C(C7=CC=C(C=C7)O)O)O)C(C)O)C)O)O)O
|
||||
| InChI |
1S/C58H73N7O17/c1-5-6-7-24-82-40-22-18-35(19-23-40)33-10-8-32(9-11-33)34-12-14-37(15-13-34)51(74)59-41-26-43(70)54(77)63-56(79)47-48(71)29(2)27-65(47)58(81)45(31(4)67)61-55(78)46(50(73)49(72)36-16-20-38(68)21-17-36)62-53(76)42-25-39(69)28-64(42)57(80)44(30(3)66)60-52(41)75/h8-23,29-31,39,41-50,54,66-73,77H,5-7,24-28H2,1-4H3,(H,59,74)(H,60,75)(H,61,78)(H,62,76)(H,63,79)/t29-,30+,31+,39+,41-,42-,43+,44-,45-,46-,47-,48-,49-,50-,54+/m0/s1
|
||||
| InChIKey |
JHVAMHSQVVQIOT-MFAJLEFUSA-N
|
||||
| CAS Number |
CAS 166663-25-8
|
||||
| ChEBI ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Voriconazole | Aspergillosis | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Up-regulation | Expression | CYP51A1 | Molecule Info |
Pathway MAP
|
|
| In-vitro Model | Aspergillus fumigatus | Microorganism model | Aspergillus fumigatus | |||
| Experimental
Result(s) |
The combination demonstrated synergy against the VCZ-R clinical isolate as well. | |||||
| Posaconazole | Aspergillosis | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [3] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Aspergillus fumigatus | Microorganism model | Aspergillus fumigatus | |||
| In-vivo Model | Six-week-old female Swiss mice were immunosuppressed with a combination of cyclophosphamide and cortisone acetate. | |||||
| Experimental
Result(s) |
The PSC and AFG combination therapy was highly effective and should be evaluated in larger-scale experiments. | |||||
| Amphotericin B | Fungal infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Experimental
Result(s) |
Anidulafungin with amphotericin B results in a synergistic effect against C. albicans and C. glabrata biofilms at serum concentrations of the drugs, but showed no interaction against C. tropicalis and C. parapsilosis complex. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Fungal 1,3-beta-glucan synthase (Fung GSC2) | Molecule Info | [5] | |
| KEGG Pathway | Starch and sucrose metabolism | Click to Show/Hide | ||
| 2 | MAPK signaling pathway - yeast | |||


