Drug Combination Details
| General Information of the Combination (ID: C80432) | |||||
|---|---|---|---|---|---|
| Name | Genistein NP Info | + | Terazosin Drug Info | ||
| Structure |
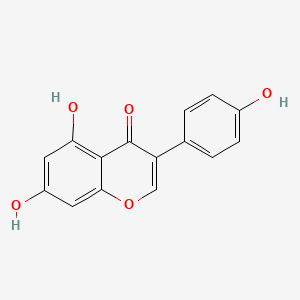
|
+ |
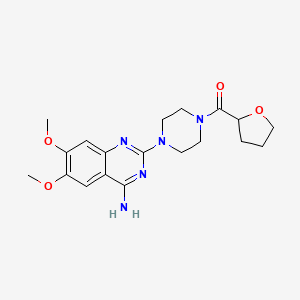
|
||
| Disease |
Prostate cancer
[ICD-11: 2C82]
|
Investigative | [1] | ||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. Enhancing Drug Efficacy by This Combination | ||||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Experiment 1 Reporting the Effect of This Combination | [1] | |||||
| Molecule(s)
Regulation |
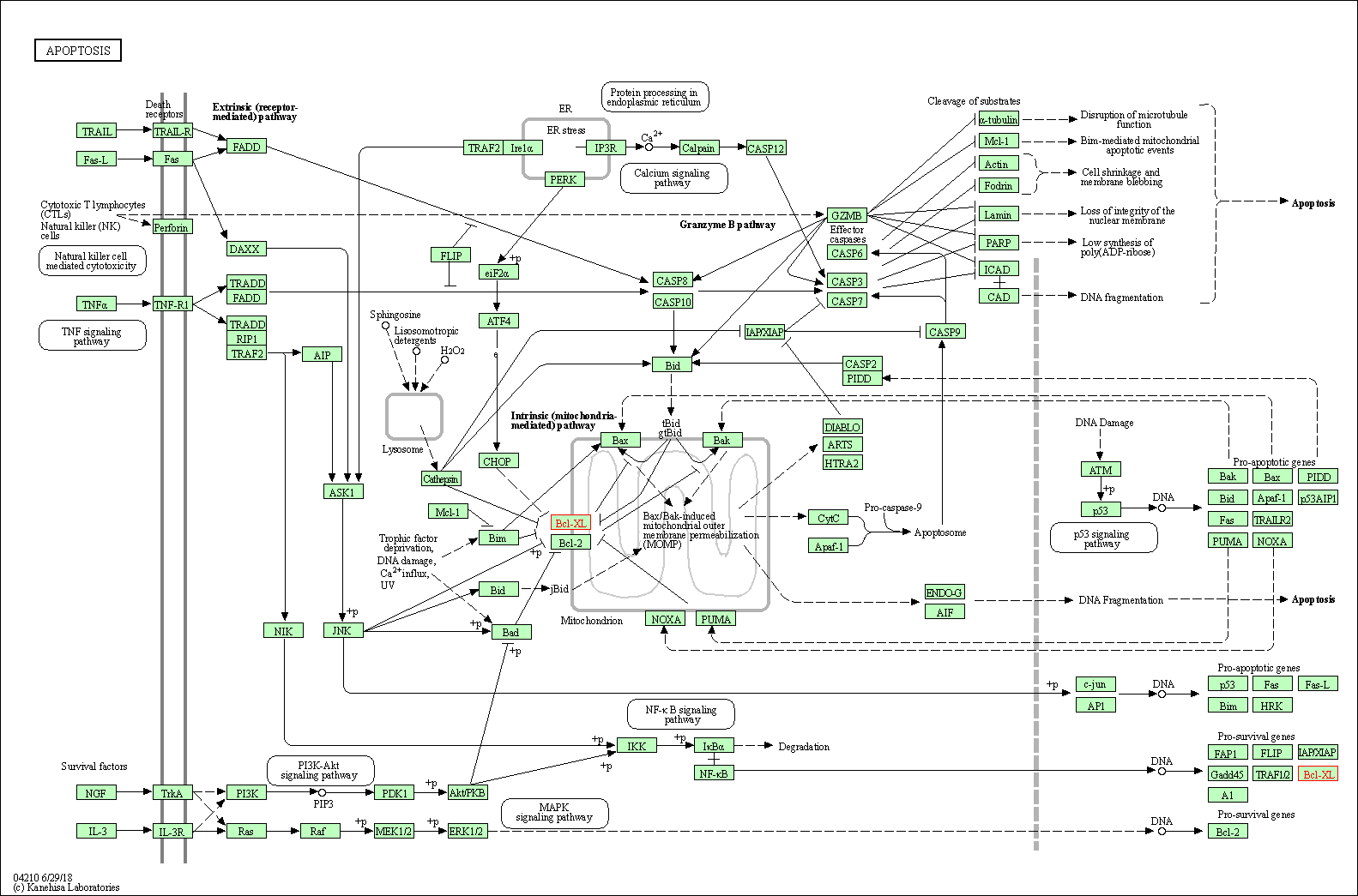
Down-regulation | Expression | BCL-xL | Molecule Info |
Pathway MAP
|
|
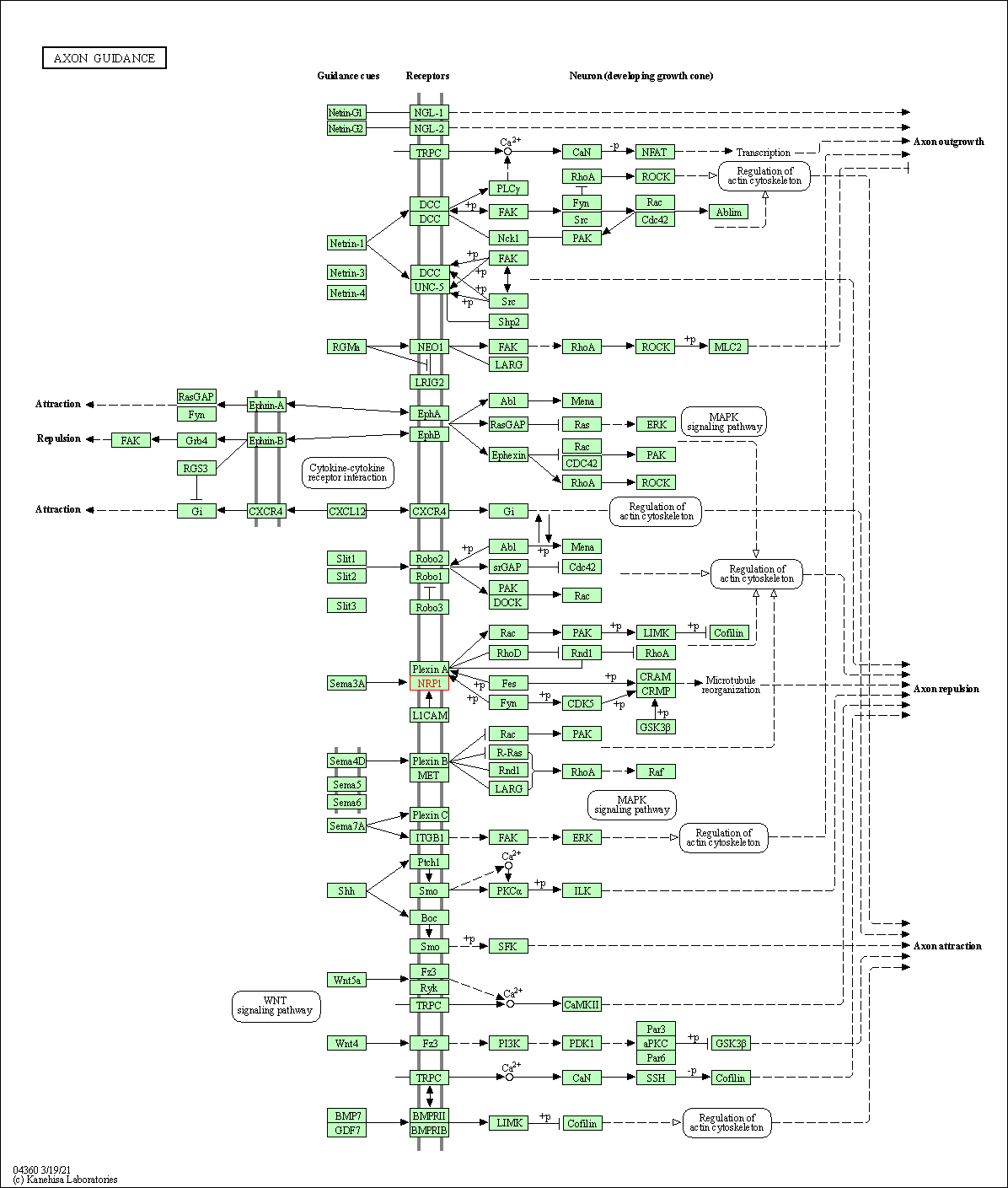
| Down-regulation | Expression | NRP1 | Molecule Info |
Pathway MAP
|
||
| In-vitro Model | DU145 | CVCL_0105 | Prostate carcinoma | Homo sapiens | ||
| Experimental
Result(s) |
The terazosin/genistein combination was more effective in inhibiting cell growth and VEGF expression as well as inducing apoptosis of the metastatic, androgen-independent prostate cancer cell line, DU-145, than either alone. | |||||
| References | ||||
|---|---|---|---|---|
| Reference 1 | Combined effects of terazosin and genistein on a metastatic, hormone-independent human prostate cancer cell line. Cancer Lett. 2009 Apr 8;276(1):14-20. | |||