Drug Details
| General Information of the Drug (ID: DR2270) | ||||
|---|---|---|---|---|
| Name |
Niclosamide
|
|||
| Synonyms |
niclosamide; 50-65-7; 5-Chloro-N-(2-chloro-4-nitrophenyl)-2-hydroxybenzamide; Niclocide; Bayluscid; Phenasal; Tredemine; Fenasal; Yomesan; 2',5-Dichloro-4'-nitrosalicylanilide; Dichlosale; Helmiantin; Atenase; Cestocid; Devermin; Devermine; Iomesan; Iomezan; Mansonil; Radeverm; Sagimid; Vermitid; Lintex; Nasemo; Sulqui; Mato; Fedal-Telmin; Bayer 73; Zestocarp; Bayer 2353; BAY 2353; Benzamide, 5-chloro-N-(2-chloro-4-nitrophenyl)-2-hydroxy-; Chemagro 2353; C13H8Cl2N2O4; 5-Chloro-2'-chloro-4'-nitrosalicylanilide; nicolsamide; HL 2447; WR 46234; Salicylanilide, 2',5-dichloro-4'-nitro-; UNII-8KK8CQ2K8G; ENT 25823; 2-Hydroxy-5-chloro-N-(2-chloro-4-nitrophenyl)benzamide; N-(2-Chloro-4-nitrophenyl)-5-chlorosalicylamide; 2-Chloro-4-nitrophenylamide-6-chlorosalicylic acid; 2',5-Dichlor-4'-nitro-salizylsaeureanilid; 5-Chloro-N-(2'-chloro-4'-nitrophenyl)salicylamide; 8KK8CQ2K8G; SR 73; Radewerm; N-(2'-Chlor-4'-nitrophenyl)-5-chlorsalicylamid; MFCD00057597; Niclosamide Anhydrous; NSC-178296; Niclosamide, 97+%; CAS-50-65-7; NCGC00015735-07; DSSTox_CID_20362; DSSTox_RID_79485; DSSTox_GSID_40362; N-(2'-Chloro-4'-nitrophenyl)-5-chlorosalicylamide; Niclosamida; Niclosamidum; Cestocide; Niclosamidum [INN-Latin]; Niclosamida [INN-Spanish]; Nitrophenyl chlorsalicylamide; Niclosamide [USAN:INN:BAN]; CCRIS 3437; HSDB 1572; SR-01000076024; Niclosamide [BSI:ISO]; EINECS 200-056-8; NSC 178296; BRN 2820605; Yomensan; AI3-25823; 5-Chlorosalicyloyl-(o-chloro-p-nitranilide); B 2353; Niclocide (TN); Prestwick_354; 2',5-Dichlor-4'-nitro-salizylsaeureanilid [German]; Mollutox (Salt/Mix); N-(2'-Chlor-4'-nitrophenyl)-5-chlorsalicylamid [German]; Spectrum_000239; Niclosamide (Niclocide); Clonitralid (Salt/Mix); Clonitralide (Salt/Mix); Prestwick0_000040; Prestwick1_000040; Prestwick2_000040; Prestwick3_000040; Spectrum2_001183; Spectrum3_000667; Spectrum4_000196; Spectrum5_001083; Lopac-N-3510; Niclosamide (USAN/INN); CHEMBL1448; Lopac0_000866; Oprea1_259151; SCHEMBL67182; BSPBio_000139; BSPBio_002333; KBioGR_000771; KBioSS_000719; MLS002154181; DivK1c_000709; SPECTRUM1503265; SPBio_001225; SPBio_002060; BPBio1_000153; CHEBI:7553; GTPL8494; DTXSID7040362; SCHEMBL18563900; WLN: WNR CG DMVR BQ EG; BDBM11242; HMS502D11; KBio1_000709; KBio2_000719; KBio2_003287; KBio2_005855; KBio3_001553; 5-Chloro-N-(2-chloro-4-nitro-phenyl)-2-hydroxy-benzamide; Niclosamide - CAS 50-65-7; NINDS_000709; HMS1568G21; HMS2093A21; HMS2095G21; HMS2231H06; HMS3262N13; HMS3373P08; HMS3712G21; KUC107299N; Pharmakon1600-01503265; BAY-2353; BCP22958; HY-B0497; ZINC3874496; Tox21_110209; Tox21_300749; Tox21_500866; BBL004110; CCG-39641; NSC178296; NSC758440; s3030; Salicylanilide,5-dichloro-4'-nitro-; STK396676; 2,5-dichloro-4-nitrosalicylanilide ?; AKOS003589004; Tox21_110209_1; BCP9000068; DB06803; KS-5210; LP00866; MCULE-9985514311; NSC-758440; SB19414; SDCCGSBI-0050841.P004; 2',5'-Dichloro-4'-nitrosalicylanilide; IDI1_000709; SMP2_000228; 5-chloro-N-(2-chloro-4-nitrophenyl)-; NCGC00015735-01; NCGC00015735-02; NCGC00015735-03; NCGC00015735-04; NCGC00015735-05; NCGC00015735-06; NCGC00015735-08; NCGC00015735-09; NCGC00015735-11; NCGC00015735-12; NCGC00015735-24; NCGC00094190-01; NCGC00094190-02; NCGC00094190-03; NCGC00094190-04; NCGC00254654-01; NCGC00261551-01; AK173020; I012; KSC-18-157-2; SMR000058390; Niclosamide 100 microg/mL in Acetonitrile; SBI-0050841.P003; AB0013423; DB-051812; VU0243604; AB00052340; EU-0100866; FT-0603220; ST50408862; T7903; EN300-92958; D00436; N 3510; AB00052340_08; AB00052340_09; A828227; Q418523; Q-201469; SR-01000076024-1; SR-01000076024-3; SR-01000076024-6; BRD-K35960502-001-06-9; BRD-K35960502-001-11-9; Z57902203; 5-Chloro-N-(2-chloro-4-nitrophenyl) -2-hydroxybenzamide; 5-Chloro-N-(2-chloro-4-nitrophenyl)-2-hydroxy-benzamide; (5-chloro-2-hydroxyphenyl)-N-(2-chloro-4-nitrophenyl)carboxamide; 5-chloranyl-N-(2-chloranyl-4-nitro-phenyl)-2-oxidanyl-benzamide; Niclosamide (anhydrous), European Pharmacopoeia (EP) Reference Standard
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Cestodes infection [ICD-11: 1F70-1F76] | Approved | [1] | |
| Structure |
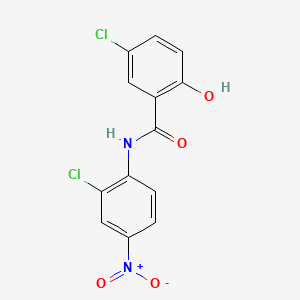
|
Click to Download Mol2D MOL |
||
| ADMET Property |
BDDCS Class
Biopharmaceutics Drug Disposition Classification System (BDDCS) Class 4: low solubility and low permeability
MRTD
The Maximum Recommended Therapeutic Dose (MRTD) of drug that ensured maximising efficacy and moderate side effect is 87.34 micromolar/kg/day
Water Solubility
The ability of drug to dissolve in water is measured as 0.013 mg/mL
Click to Show/Hide
|
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C13H8Cl2N2O4
|
|||
| PubChem CID | ||||
| Canonical SMILES |
C1=CC(=C(C=C1[N+](=O)[O-])Cl)NC(=O)C2=C(C=CC(=C2)Cl)O
|
|||
| InChI |
1S/C13H8Cl2N2O4/c14-7-1-4-12(18)9(5-7)13(19)16-11-3-2-8(17(20)21)6-10(11)15/h1-6,18H,(H,16,19)
|
|||
| InChIKey |
RJMUSRYZPJIFPJ-UHFFFAOYSA-N
|
|||
| CAS Number |
CAS 50-65-7
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Reverse the Resistance of This Drug | ||||||
| Colistin | Bacillus polymyxa | Click to Show/Hide the Molecular Data of This NP | ||||
| Reversing Drug Resistance | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Biological
Regulation |
Increase | Zeta potential | ||||
| In-vitro Model | Acinetobacter baumannii | Microorganism model | Acinetobacter baumannii | |||
| Klebsiella pneumoniae | Microorganism model | Klebsiella pneumoniae | ||||
| Experimental
Result(s) |
Niclosamide has potentiated the effect of colistin against Col-S and Col-R A. | |||||

