Drug Details
| General Information of the Drug (ID: DR5153) | ||||
|---|---|---|---|---|
| Name |
Spironolactone
|
|||
| Synonyms |
spironolactone; 52-01-7; Aldactone; Spirolactone; Verospiron; Euteberol; Spirolang; Verospirone; Spiroctan; Acelat; Aldactone A; Spironocompren; Melarcon; Spiresis; Spiridon; Uractone; Urusonin; Alderon; Spirone; Xenalon; Dira; Spiro-Tablinen; Spironolactone A; spironolattone; Aquareduct; Aldopur; Espironolactona; Spironolactonum; Aldace; Almatol; Deverol; Altex; Berlactone; SC 9420; SC-9420; UNII-27O7W4T232; MFCD00082250; CHEMBL1393; C24H32O4S; CHEBI:9241; Spiro(17H-cyclopenta(a)phenauthrene-17,2'-(3'H)-furan); Spironolattone [DCIT]; 27O7W4T232; NSC-150399; S-[(7R,8R,9S,10R,13S,14S,17R)-10,13-dimethyl-3,5'-dioxospiro[2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthrene-17,2'-oxolane]-7-yl] ethanethioate; Spironolactone, 99%; Aldactide; Spiractin; Spironolactonum [INN-Latin]; Espironolactona [INN-Spanish]; DSSTox_CID_14186; DSSTox_RID_79120; DSSTox_GSID_34186; SC 15983; (1'S,2R,2'R,9'R,10'R,11'S,15'S)-9'-(acetylsulfanyl)-2',15'-dimethylspiro[oxolane-2,14'-tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan]-6'-ene-5,5'-dione; S-[(7R,8R,9S,10R,13S,14S,17R)-10,13-dimethyl-3,5'-dioxo-1,2,3,4',5',6,7,8,9,10,11,12,13,14,15,16-hexadecahydro-3'H-spiro[cyclopenta[a]phenanthrene-17,2'-furan]-7-yl] ethanethioate; Spiro[17H-cyclopenta[a]phenauthrene-17,2'-(3'H)-furan]; Diatense; Aldactone (TN); 17-Hydroxy-7-alpha-mercapto-3-oxo-17-alpha-pregn-4-ene-21-carboxylic acid-gamma-lactone-7-acetate; SMR000471892; HSDB 3184; EINECS 200-133-6; NSC 150399; BRN 0057767; spirotone; Spiranolactone; Carospir; 2oax; 3vhu; CAS-52-01-7; NCGC00015948-02; 3-(3-Keto-7.alpha.-acetylthio-17.beta.-hydroxy-4-androsten-17.alpha.-yl)propionic acid lactone; 7-.alpha.-(acetylthio)-17-.alpha.-hydroxy-3-oxopregn-4-ene-21-carboxylic acid, .gamma.-lactone; 7-alpha-Acetylthio-3-oxo-17-alpha-pregn-4-ene-21,17-beta-carbolactone; S-((7R,8R,9S,10R,13S,14S,17R)-10,13-dimethyl-3,5'-dioxo-1,2,3,4',5',6,7,8,9,10,11,12,13,14,15,16-hexadecahydro-3'H-spiro[cyclopenta[a]phenanthrene-17,2'-furan]-7-yl) ethanethioate; Spironolactone [USP:INN:BAN:JAN]; Pregn-4-ene-21-carboxylic acid, 7-(acetylthio)-17-hydroxy-3-oxo-, gamma-lactone, (7alpha,17alpha)-; Prestwick0_000128; Prestwick1_000128; Prestwick2_000128; Prestwick3_000128; 17-Hydroxy-7alpha-mercapto-3-oxo-17alpha-pregn-4-ene-21-carboxylic acid, gamma-lactone acetate; 3'-(3-Oxo-7-alpha-acetylthio-17-beta-hydroxyandrost-4-en-17-beta-yl)propionic acid lactone; 3-(3-Keto-7-alpha-acetylthio-17-beta-hydroxy-4-androsten-17-alpha-yl)propionic acid lactone; BIDD:PXR0071; SCHEMBL20939; BSPBio_000176; 17alpha-Pregn-4-ene-21-carboxylic acid, 17-hydroxy-7alpha-mercapto-3-oxo-, gamma-lactone, acetate; 4-18-00-01601 (Beilstein Handbook Reference); MLS001074672; MLS001333253; MLS001333254; MLS002153245; MLS002207058; MLS002548846; SPBio_002115; BPBio1_000194; GTPL2875; DTXSID6034186; Spironolactone (JP17/USP/INN); HMS1568I18; HMS2090N21; HMS2095I18; HMS2236E06; HMS3259G11; HMS3712I18; ACT02596; AMY40521; HY-B0561; Spironolactone, 97.0-103.0%; ZINC3861599; Tox21_113047; Tox21_302154; 7alpha-(acetylsulfanyl)-3-oxo-17alpha-pregn-4-ene-21,17-carbolactone; BDBM50228080; NSC150399; s4054; 4-Pregnen-21-oic acid-17alpha-ol-3-one-7alpha-thiol gamma-lactone 7-acetate; AKOS015896401; AC-4214; CCG-220128; DB00421; KS-5234; MCULE-4394091100; NC00482; NCGC00164397-01; NCGC00164397-02; NCGC00164397-03; NCGC00164397-05; NCGC00255229-01; 17-alpha-Pregn-4-ene-21-carboxylic acid, 17-hydroxy-7-alpha-mercapto-3-oxo-, gamma-lactone acetate; AK546740; CPD000471892; H490; Spironolactone 1.0 mg/ml in Acetonitrile; AB00513806; S0260; 7080-EP2269989A1; 7080-EP2270011A1; 7080-EP2272841A1; 7080-EP2295406A1; 7080-EP2298772A1; 7080-EP2298776A1; 7080-EP2301936A1; 7080-EP2308562A2; 7080-EP2308839A1; C07310; D00443; AB01275520-01; AB01275520_02; 082S250; Q422188; SR-01000765419; SR-05000000452; Q-201737; SR-01000765419-2; SR-05000000452-2; BRD-K90027355-001-03-4; BRD-K90027355-001-19-0; Z1551900341; Spironolactone, European Pharmacopoeia (EP) Reference Standard; WLN: L E5 B666 FX OV MUTJ A1 E1 KSV1 F-& CT5VOXTJ; 17.alpha.-Pregn-4-ene-21-carboxylic acid, .gamma.-lactone acetate; Spironolactone, United States Pharmacopeia (USP) Reference Standard; 17.alpha.-Pregn-4-ene-21-carboxylic acid, .gamma.-lactone, acetate; Pregn-4-ene-21-carboxylic acid, .gamma.-lactone, (7.alpha.,17.alpha.)-; (7?,17?)-7-(Acetylthio)-17-hydroxy-3-oxopregn-4-ene-21-carboxylic acid ?-lactone; 7alpha-(Acetylthio)-17alpha-hydroxy-3-oxopregn-4-ene-21-carboxylic acid gamma-lactone; Spironolactone for system suitability, European Pharmacopoeia (EP) Reference Standard; 17-HYDROXY-7ALPHA-MERCAPTO-3-OXO-17ALPHA-PREGN-4-ENE-21- CARBOXYLIC ACID; GAMMA-LACTONE ACETATE; 2'',15''-dimethyl-5,5''-dioxo-(9''R)-spiro[tetrahydrofuran-2,14''-tetracyclo[8.7.0.02,7.011,15]heptadec-6''-ene]-9-yl ethanethioate; 2'',15''-dimethyl-5,5''-dioxospiro[tetrahydrofuran-2,14''-tetracyclo[8.7.0.02,7.011,15]heptadec-6''-ene]-9-yl ethanethioate; 2'',15''-dimethyl-5,5''-dioxospiro[tetrahydrofuran-2,14''-tetracyclo[8.7.0.02,7.011,15]heptadec-6''-ene]-9-yl ethanethioate(Spiranolactone); 7-alpha-(acetylthio)-17-alpha-hydroxy-3-oxopregn-4-ene-21-carboxylic acid, gamma-lactone (9CI); S-(2''R,7R,8R,9S,10R,13S,14S)-10,13-dimethyl-3,5''-dioxo-1,2,3,4'',5'',6,7,8,9,10,11,12,13,14,15,16-hexadecahydro-3''H-spiro[cyclopenta[a]phenanthrene-17,2''-furan]-7-yl ethanethioate
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Heart failure [ICD-11: BD10] | Approved | [1] | |
| Structure |
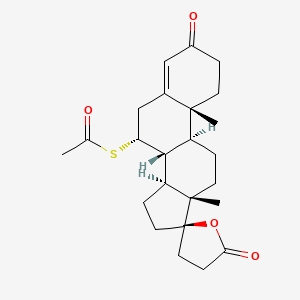
|
Click to Download Mol2D MOL |
||
| ADMET Property |
BDDCS Class
Biopharmaceutics Drug Disposition Classification System (BDDCS) Class 2: low solubility and high permeability
Elimination
0.5% of drug is excreted from urine in the unchanged form
MRTD
The Maximum Recommended Therapeutic Dose (MRTD) of drug that ensured maximising efficacy and moderate side effect is 13.71714 micromolar/kg/day
Water Solubility
The ability of drug to dissolve in water is measured as 0.022 mg/mL
Click to Show/Hide
|
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C24H32O4S
|
|||
| PubChem CID | ||||
| Canonical SMILES |
CC(=O)SC1CC2=CC(=O)CCC2(C3C1C4CCC5(C4(CC3)C)CCC(=O)O5)C
|
|||
| InChI |
1S/C24H32O4S/c1-14(25)29-19-13-15-12-16(26)4-8-22(15,2)17-5-9-23(3)18(21(17)19)6-10-24(23)11-7-20(27)28-24/h12,17-19,21H,4-11,13H2,1-3H3/t17-,18-,19+,21+,22-,23-,24+/m0/s1
|
|||
| InChIKey |
LXMSZDCAJNLERA-ZHYRCANASA-N
|
|||
| CAS Number |
CAS 52-01-7
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Vitamin D | Saccharomyces cerevisiae | Click to Show/Hide the Molecular Data of This NP | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
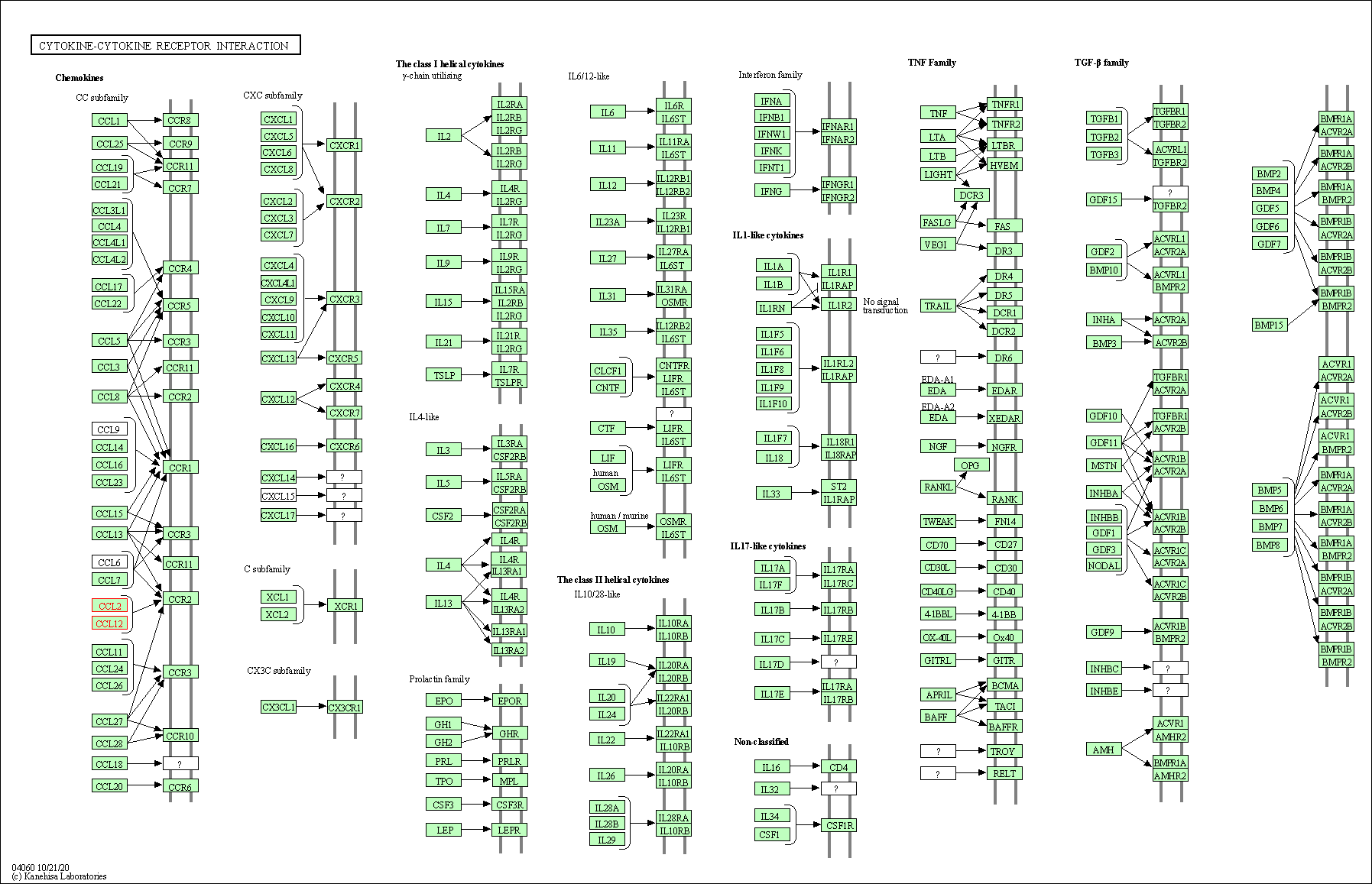
Down-regulation | Expression | CCL2 | Molecule Info |
Pathway MAP
|
|
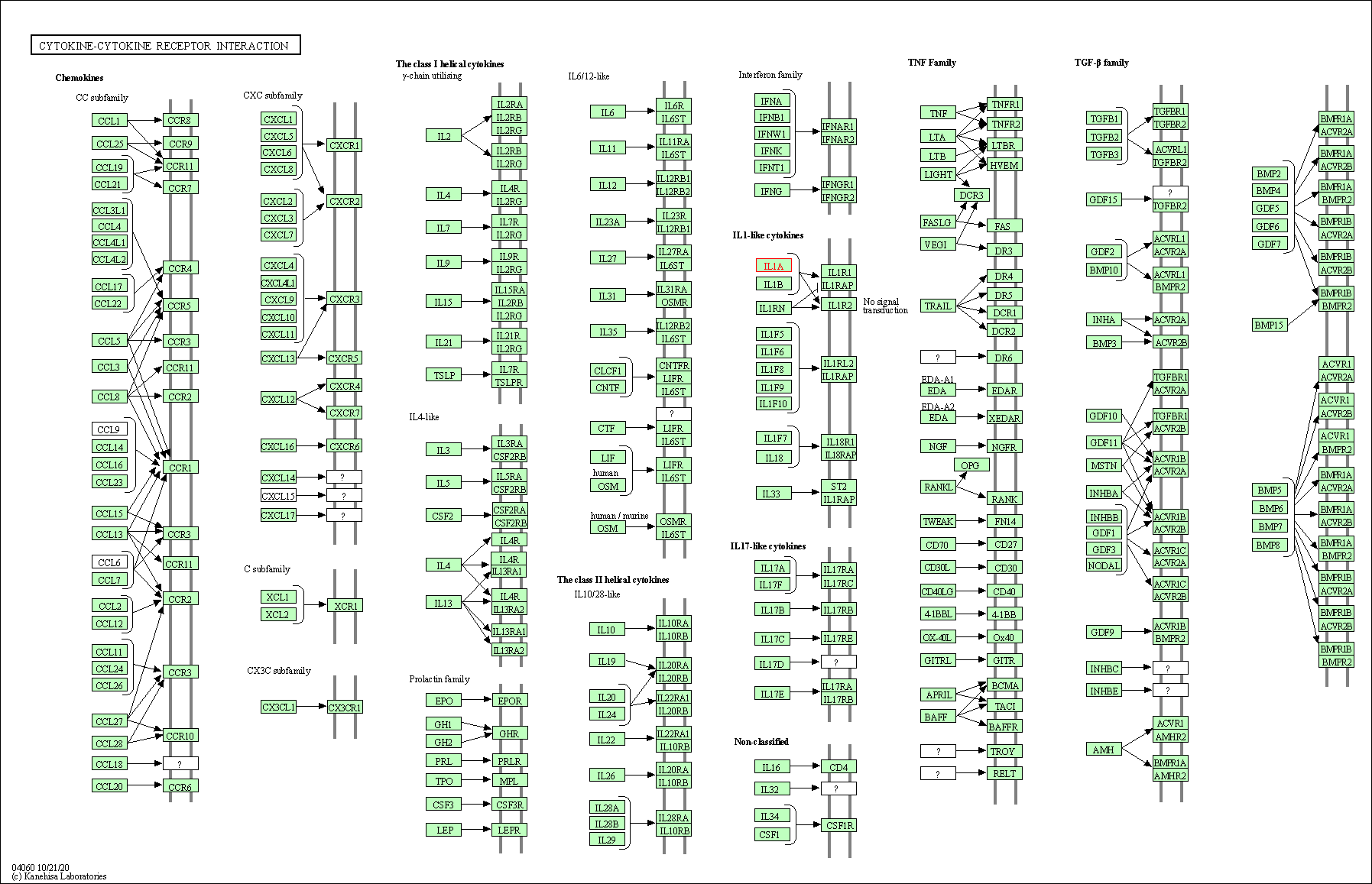
| Down-regulation | Expression | IL1A | Molecule Info |
Pathway MAP
|
||
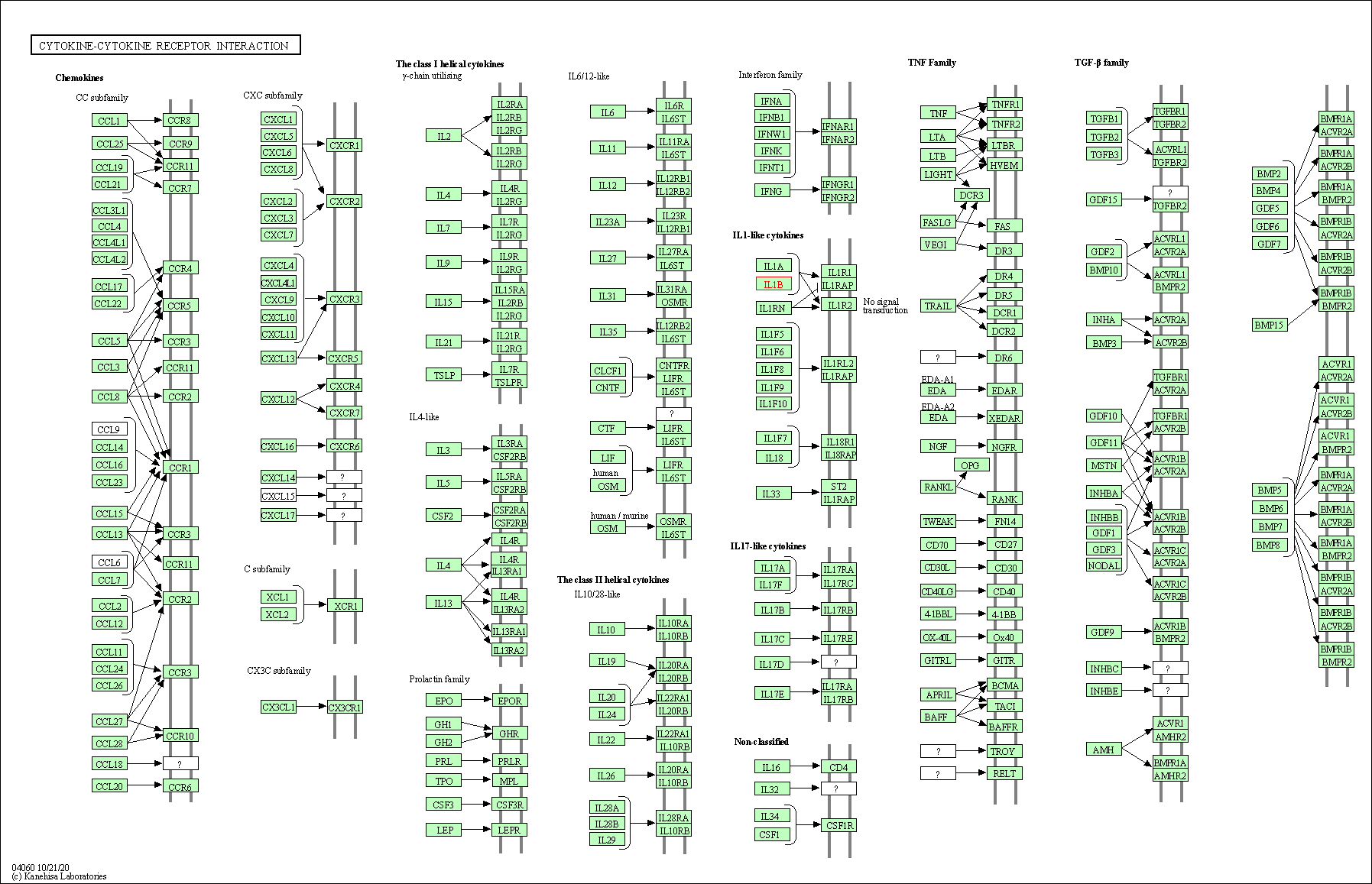
| Down-regulation | Expression | IL1B | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | MMP-9 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | NOS2 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | PDK1 | Molecule Info |
Pathway MAP
|
||
| Biological
Regulation |
Increase | M2/M1 ratio | ||||
| In-vitro Model | RAW-Dual | CVCL_A7ZK | Mouse leukemia | Mus musculus | ||
| In-vivo Model | Six- to eight-week-old C57BL/6J female mice were used in this study. | |||||
| Experimental
Result(s) |
VD3 and SP may constitute an effective treatment regimen to improve wound healing after NM or other skin chemical injury. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Mineralocorticoid receptor (MR) | Molecule Info | [3] | |
| KEGG Pathway | Aldosterone-regulated sodium reabsorption | Click to Show/Hide | ||
| Pathwhiz Pathway | Kidney Function | Click to Show/Hide | ||
| WikiPathways | ACE Inhibitor Pathway | Click to Show/Hide | ||