Drug Details
| General Information of the Drug (ID: DR5724) | ||||
|---|---|---|---|---|
| Name |
Ribavirin
|
|||
| Synonyms |
ribavirin; 36791-04-5; Tribavirin; Rebetol; Virazole; Copegus; Vilona; Ribavirine; Viramid; Ribamide; Ribamidil; Ribasphere; Ribavirina; Ribavirinum; Ribavirine [INN-French]; Ribavirinum [INN-Latin]; ICN-1229; Ribavirina [INN-Spanish]; 1-beta-D-Ribofuranosyl-1,2,4-triazole-3-carboxamide; 1-beta-D-Ribofuranosyl-1H-1,2,4-triazole-3-carboxamide; RTCA; RBV; SCH 18908; UNII-49717AWG6K; Ribamidyl; Ribavirin (Copegus); 1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-1H-1,2,4-triazole-3-carboxamide; 1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-1H-1,2,4-triazole-3-carboxamide; 1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,2,4-triazole-3-carboxamide; MFCD00058564; Rebetron; Varazid; Ribavirin Capsules; CHEBI:63580; 49717AWG6K; Ribavirin, 98%; 1-(beta-D-ribofuranosyl)-1H-1,2,4-triazole-3-carboxamide; Rebretron; Virazid; Virazide; Ribav; DSSTox_CID_3557; DSSTox_RID_77081; DSSTox_GSID_23557; RTC; 1-(beta-D-Ribofuranosyl)-1,2,4-triazole-3-carboxamide; Cotronak; Ravanex; Ribacine; NSC-163039; C-Virin; DRG-0028; Ribasphere (TN); Virazole (TN); SMR000058315; Copegus (TN); Rebetol (TN); HSDB 6513; SR-01000076112; BRN 0892462; Ribavirine;; NSC163039; Ribavirin [USAN:USP:INN:BAN]; Ribavirin,(S); NCGC00015904-02; Ribavirin, antiviral; CAS-36791-04-5; RG-964; PubChem14187; Ro-20-9963; Spectrum_001826; 1H-1,2,4-Triazole-3-carboxamide, 1-beta-D-ribofuranosyl-; 4pb1; ribofluranosyl carboxamide; Prestwick3_000993; Spectrum3_001876; Spectrum4_001252; Spectrum5_002075; R-964; R 9644; SCHEMBL3727; CHEMBL1643; Lopac0_001063; BSPBio_001085; BSPBio_003352; KBioGR_001804; KBioSS_002331; cid_37542; MLS000028486; MLS002222317; DivK1c_000782; SPECTRUM1503938; Ribavirin (JP17/USP/INN); BPBio1_001195; GTPL6842; 1,2,4-Triazole-3-carboxamide, 1-beta-D-ribofuranosyl-; 1-.beta.-D-Ribofuranosyl-1,2,4-triazolo-3-carboxamide; DTXSID8023557; HMS502H04; KBio1_000782; KBio2_002328; KBio2_004896; KBio2_007464; KBio3_002854; NINDS_000782; HMS2090L15; HMS2094O09; HMS2098G07; HMS2232P07; HMS3263E08; HMS3715G07; Pharmakon1600-01503938; HY-B0434; ZINC1035331; Tox21_110259; Tox21_200967; Tox21_501063; BDBM50154375; CCG-38985; NSC758650; s2504; SCH-18908; AKOS001715163; Tox21_110259_1; DB00811; GS-3572; LP01063; MCULE-9495128592; NSC-758650; SDCCGSBI-0051033.P004; IDI1_000782; SMP1_000261; NCGC00090726-01; NCGC00090726-03; NCGC00090726-04; NCGC00090726-05; NCGC00090726-06; NCGC00090726-07; NCGC00090726-08; NCGC00090726-09; NCGC00090726-12; NCGC00090726-25; NCGC00258520-01; NCGC00261748-01; 252269-50-4; AS-34178; BCP0726000138; SBI-0051033.P003; AB0068936; AB00430481; EU-0101063; R0077; EN300-59237; D00423; J10190; M-7546; AB00430481-15; AB00430481-16; AB00430481_17; AB00430481_18; 1-beta-Ribofuranosyl-1,2,4-triazole-3-carboamide; 791R045; Q421862; Ribavirin 100 microg/mL in Acetonitrile:Methanol; SR-01000721904; SR-01000076112-2; SR-01000076112-3; SR-01000076112-4; SR-01000721904-2; 1-??-D-Ribofuranosyl-1,2,4-triazole-3-carboxamide; BRD-K60369935-001-02-7; BRD-K60369935-001-18-3; SR-01000076112-11; 1-?-D-Ribofuranosyl-1H-1,2,4-triazole-3-carboxamide; Ribavirin, British Pharmacopoeia (BP) Reference Standard; Z1522567185; 1-(beta -D-ribofuranosyl)-1,2,4-triazole-3-carboxamide; Ribavirin, European Pharmacopoeia (EP) Reference Standard; Ribavirin, United States Pharmacopeia (USP) Reference Standard; Ribavirin, Pharmaceutical Secondary Standard; Certified Reference Material; 1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-1,2,4-triazole-3-carboxamide
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Hepatitis C virus infection [ICD-11: 1E51] | Approved | [1] | |
| Structure |
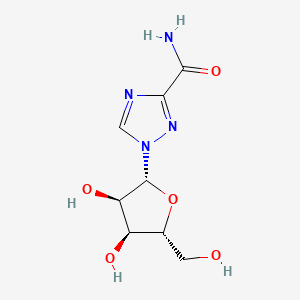
|
Click to Download Mol2D MOL |
||
| ADMET Property |
Absorption Tmax
The time to maximum plasma concentration (Tmax) is 2 h
BDDCS Class
Biopharmaceutics Drug Disposition Classification System (BDDCS) Class 1: high solubility and high permeability
Bioavailability
60% of drug becomes completely available to its intended biological destination(s)
Clearance
The clearance of drug is 26 L/h
Clearance
The drug present in the plasma can be removed from the body at the rate of 5.2 mL/min/kg
Elimination
After the oral administration of 600mg radiolabeled ribavirin, approximately 61% of the drug was detected in the urine and 12% was detected in the feces
Elimination
17% of drug is excreted from urine in the unchanged form
Half-life
The concentration or amount of drug in body reduced by one-half in 120 - 170 hours
Half-life
The concentration or amount of drug in body reduced by one-half in 45 hours
Metabolism
The drug is metabolized via the adenosine kinase
MRTD
The Maximum Recommended Therapeutic Dose (MRTD) of drug that ensured maximising efficacy and moderate side effect is 35.1 micromolar/kg/day
Unbound Fraction
The unbound fraction of drug in plasma is 1%
Vd
Fluid volume that would be required to contain the amount of drug present in the body at the same concentration as in the plasma 14 L/kg
Water Solubility
The ability of drug to dissolve in water is measured as 142 mg/mL
Click to Show/Hide
|
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C8H12N4O5
|
|||
| PubChem CID | ||||
| Canonical SMILES |
C1=NC(=NN1C2C(C(C(O2)CO)O)O)C(=O)N
|
|||
| InChI |
1S/C8H12N4O5/c9-6(16)7-10-2-12(11-7)8-5(15)4(14)3(1-13)17-8/h2-5,8,13-15H,1H2,(H2,9,16)/t3-,4-,5-,8-/m1/s1
|
|||
| InChIKey |
IWUCXVSUMQZMFG-AFCXAGJDSA-N
|
|||
| CAS Number |
CAS 36791-04-5
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Jerusalem artichoke + Interferon alpha-2a | Click to Show/Hide the Molecular Data of This NP | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
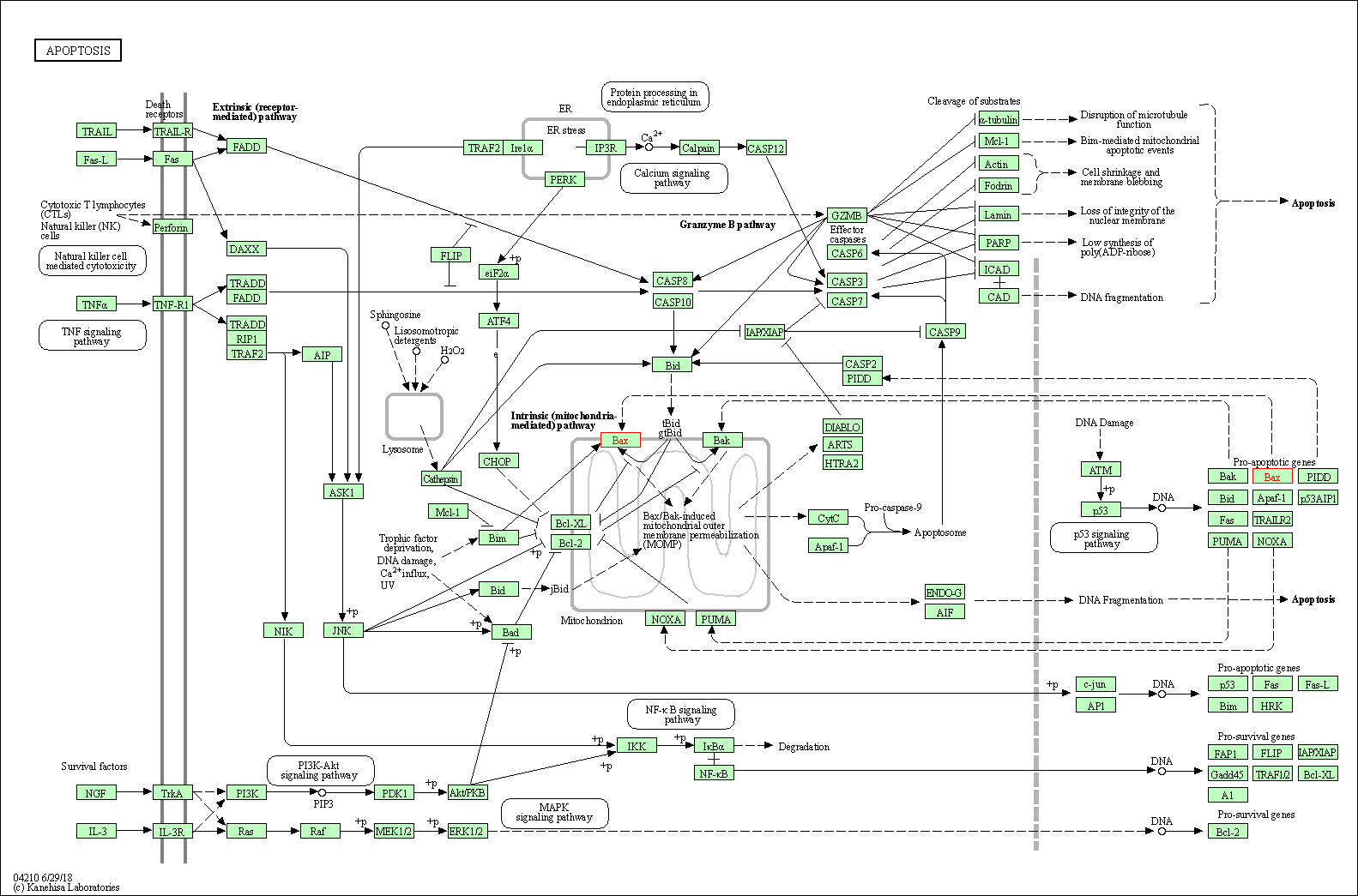
Down-regulation | Expression | BAX | Molecule Info |
Pathway MAP
|
|
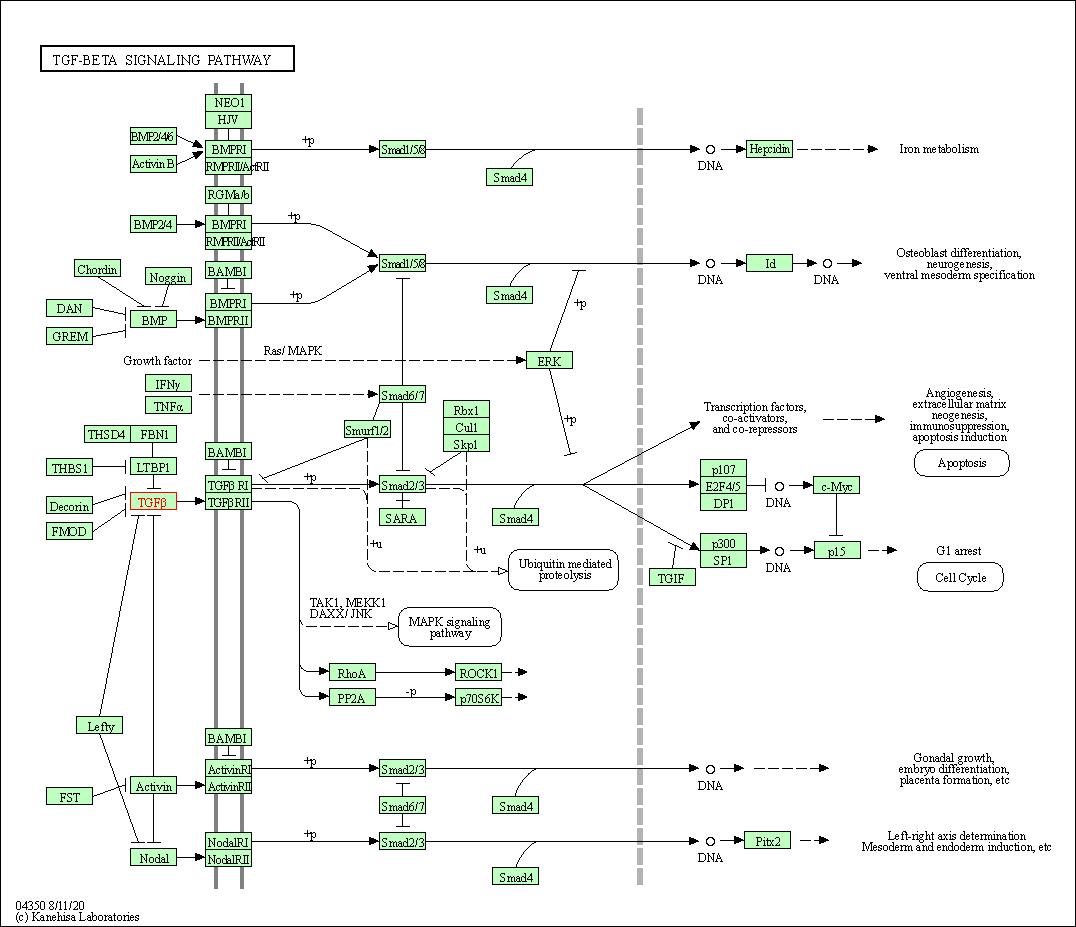
| Down-regulation | Expression | TGFB1 | Molecule Info |
Pathway MAP
|
||
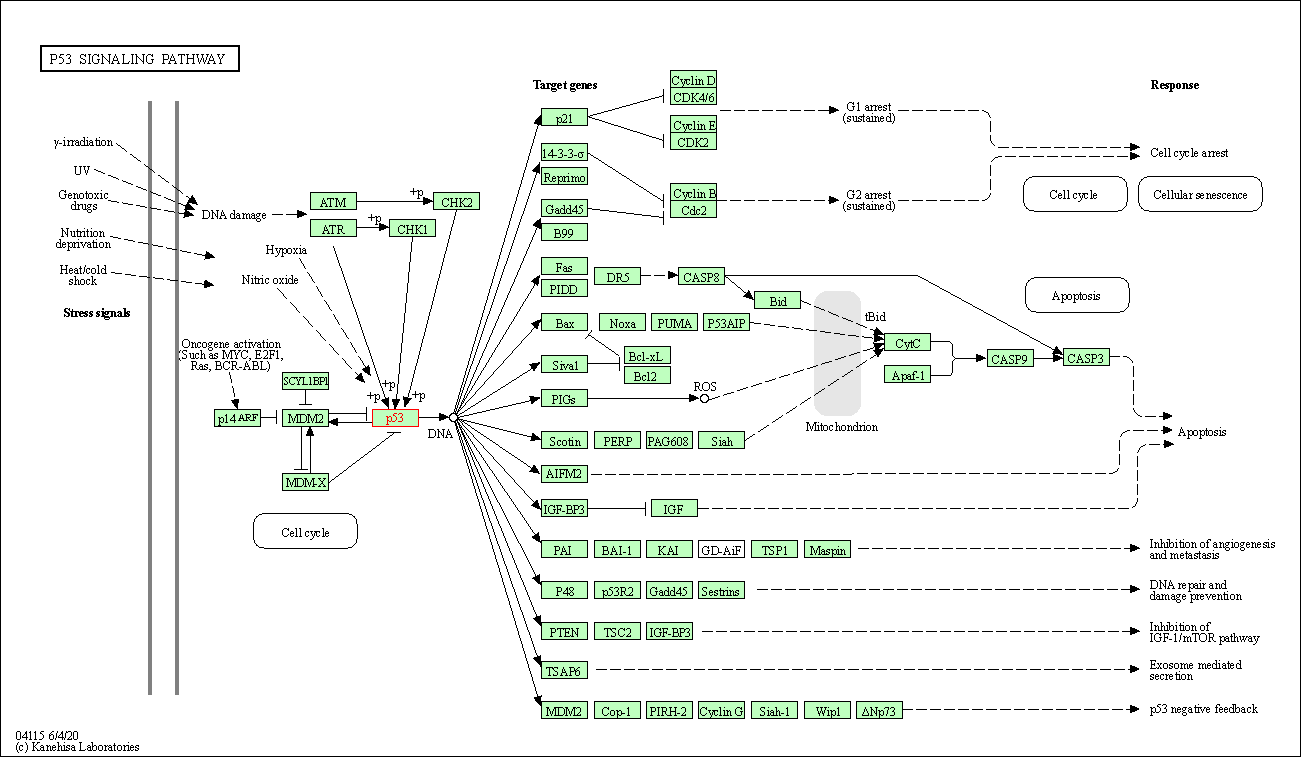
| Down-regulation | Expression | TP53 | Molecule Info |
Pathway MAP
|
||
| In-vivo Model | Male albino rats (150-200g) were used in this study. | |||||
| Experimental
Result(s) |
Addition of JAT as a supportive regimen to interferon and ribavirin effectively potentiates their anti-fibrotic effects. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | IMP dehydrogenase 1 (IMPDH1) | Molecule Info | [3] | |