Drug Details
| General Information of the Drug (ID: DR6231) | ||||
|---|---|---|---|---|
| Name |
Gilteritinib
|
|||
| Synonyms |
Gilteritinib; 1254053-43-4; ASP2215; ASP-2215; Xospata; UNII-66D92MGC8M; ASP 2215; 6-Ethyl-3-((3-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)-amino)-5-((tetrahydro-2H-pyran-4-yl)amino)pyrazine-2-carboxamide; 66D92MGC8M; Gilteritinib HCl; 1254053-43-4 (free base); 2-Pyrazinecarboxamide, 6-ethyl-3-((3-methoxy-4-(4-(4-methyl-1-piperazinyl)-1-piperidinyl)phenyl)amino)-5-((tetrahydro-2H-pyran-4-yl)amino)-; 2-Pyrazinecarboxamide, 6-ethyl-3-[[3-methoxy-4-[4-(4-methyl-1-piperazinyl)-1-piperidinyl]phenyl]amino]-5-[(tetrahydro-2H-pyran-4-yl)amino]-; 6-ethyl-3-((3-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)amino)-5-((tetrahydro-2H-pyran-4-yl)amino)pyrazine-2-carboxamide.; 6-ethyl-3-[[3-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl]amino]-5-(oxan-4-ylamino)pyrazine-2-carboxamide; 6-ethyl-3-[3-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]anilino]-5-(oxan-4-ylamino)pyrazine-2-carboxamide; Gilteritinib [USAN:INN]; Gilteritinib (ASP2215); gilteritinibum; C6F; Gilteritinib(ASP2215); Gilteritinib (USAN/INN); Gilteritinib (ASP-2215); SCHEMBL282229; GTPL8708; CHEMBL3301622; CHEBI:145372; BDBM144315; C29H44N8O3; BCP28756; EX-A2775; 3694AH; MFCD28144685; NSC787846; NSC787854; NSC788454; NSC800106; s7754; ZINC113476229; CCG-270016; CS-3885; DB12141; NSC-787846; NSC-787854; NSC-788454; NSC-800106; SB16988; NCGC00481652-01; NCGC00481652-02; AC-29030; AS-35199; HY-12432; QC-11768; DB-108103; A14411; D10709; US8969336, 547; US8969336, 577; Q27077802; 6-ethyl-3-((3-methoxy-4-(4-(4-methyl-1-piperazinyl)-1-piperidinyl)phenyl)amino)-5-((tetrahydro-2H-pyran-4-yl)amino)-2-pyrazinecarboxamide; 6-Ethyl-3-((3-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)amino)-5-((tetrahydro-2H-pyran-4-yl)amino)pyrazine-2-carboxamide; 6-ethyl-3-({3-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl}amino)-5-(tetrahydro-2H-pyran-4-ylamino)pyrazine-2-carboxamide; 6-Ethyl-3-[3-methoxy-4-[4-(4-methylpiperazine-1-yl)piperidino]anilino]-5-[(tetrahydro-2H-pyran-4-yl)amino]pyrazine-2-carboxamide; 6-ethyl-3-{3-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]anilino}-5-(tetrahydro-2H-pyran-4-ylamino)pyrazine-2-carboxamide; 6-ethyl-3-{3-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]anilino}-5-[(oxan-4-yl)amino]pyrazine-2-carboxamide
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Acute myeloid leukemia [ICD-11: 2A60] | Approved | [1] | |
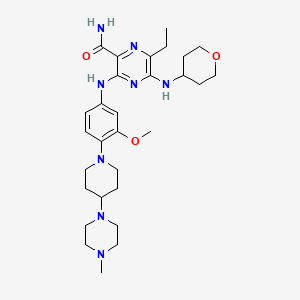
| Structure |
|
Click to Download Mol2D MOL |
||
| ADMET Property |
Absorption AUC
The area under the plot of plasma concentration (AUC) of drug is 6943 mcgh/L
Absorption Cmax
The maximum plasma concentration (Cmax) of drug is 374 mcg/L
Absorption Tmax
The time to maximum plasma concentration (Tmax) is 4-8 h
Clearance
The clearance of drug is 14.85 L/h
Elimination
From the administered dose, gilteritinib is mainly excreted in feces which represents 64.5% of the administered dose while 16.4% is recovered in urine either as the unchanged drug or as its metabolites
Half-life
The concentration or amount of drug in body reduced by one-half in 45 - 159 hours
Metabolism
The drug is metabolized via the liver
Vd
The volume of distribution (Vd) of drug is 1092 L
Click to Show/Hide
|
|||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C29H44N8O3
|
|||
| PubChem CID | ||||
| Canonical SMILES |
CCC1=C(N=C(C(=N1)C(=O)N)NC2=CC(=C(C=C2)N3CCC(CC3)N4CCN(CC4)C)OC)NC5CCOCC5
|
|||
| InChI |
1S/C29H44N8O3/c1-4-23-28(31-20-9-17-40-18-10-20)34-29(26(33-23)27(30)38)32-21-5-6-24(25(19-21)39-3)37-11-7-22(8-12-37)36-15-13-35(2)14-16-36/h5-6,19-20,22H,4,7-18H2,1-3H3,(H2,30,38)(H2,31,32,34)
|
|||
| InChIKey |
GYQYAJJFPNQOOW-UHFFFAOYSA-N
|
|||
| CAS Number |
CAS 1254053-43-4
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Arsenic trioxide | Realgar and orpiment | Click to Show/Hide the Molecular Data of This NP | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
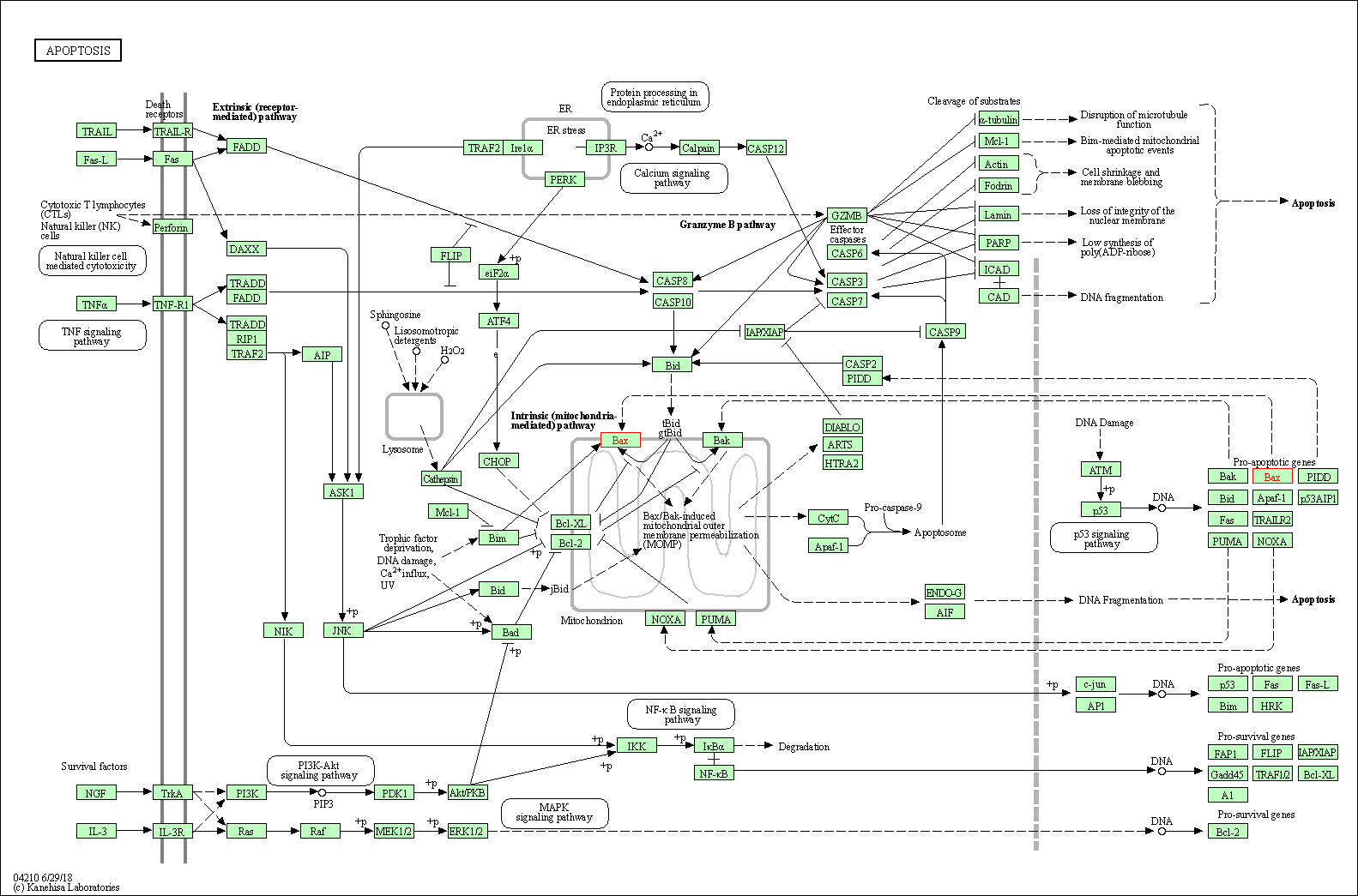
Up-regulation | Expression | BAX | Molecule Info |
Pathway MAP
|
|
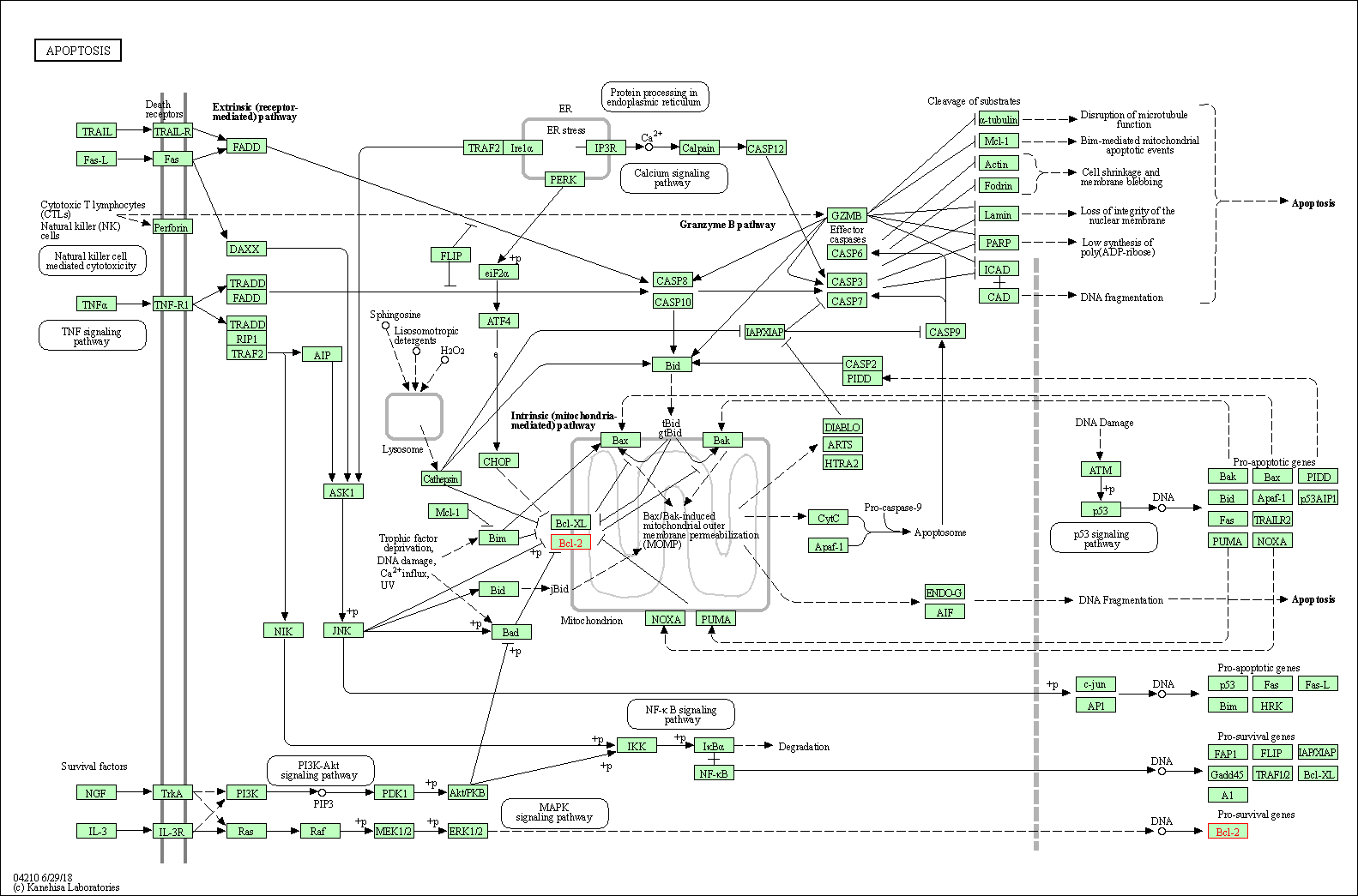
| Down-regulation | Expression | BCL-2 | Molecule Info |
Pathway MAP
|
||
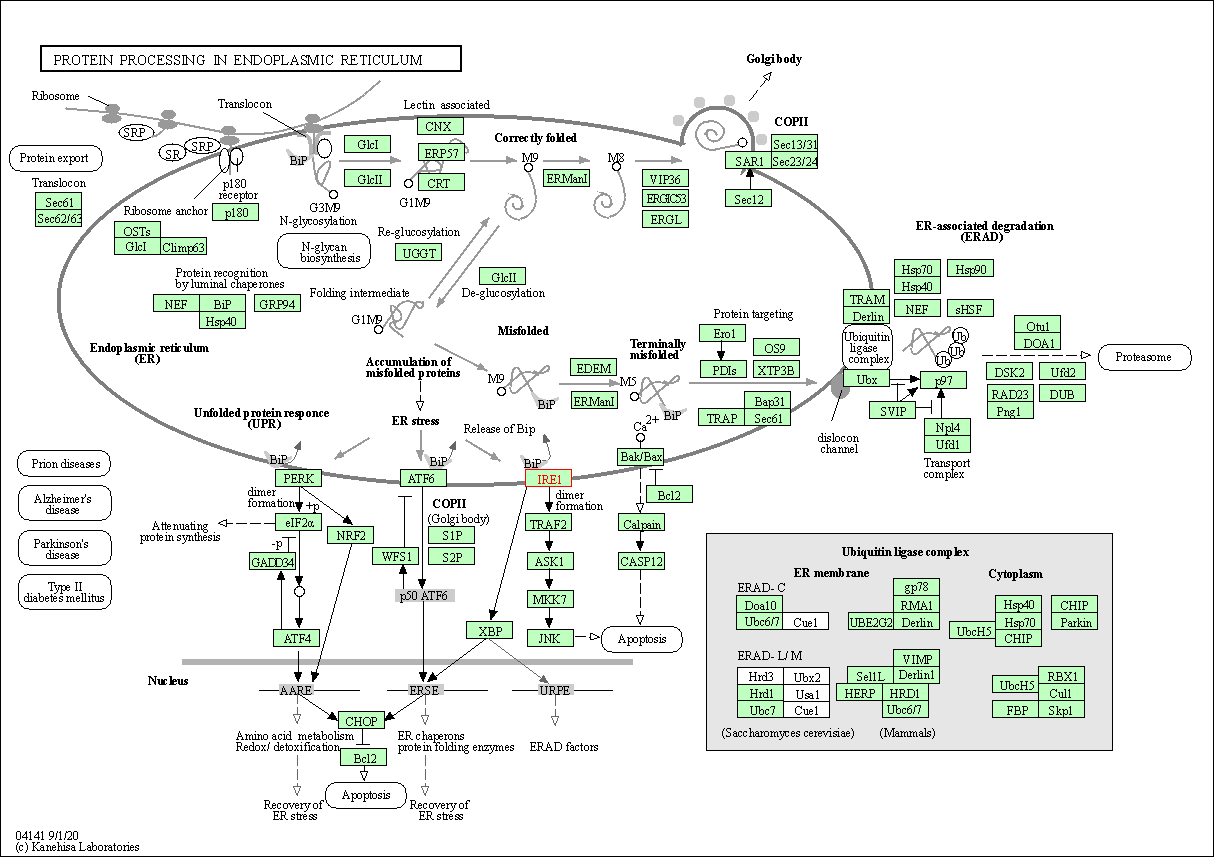
| Down-regulation | Expression | ERN1 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Phosphorylation | FLT-3 | Molecule Info |
Pathway MAP
|
||
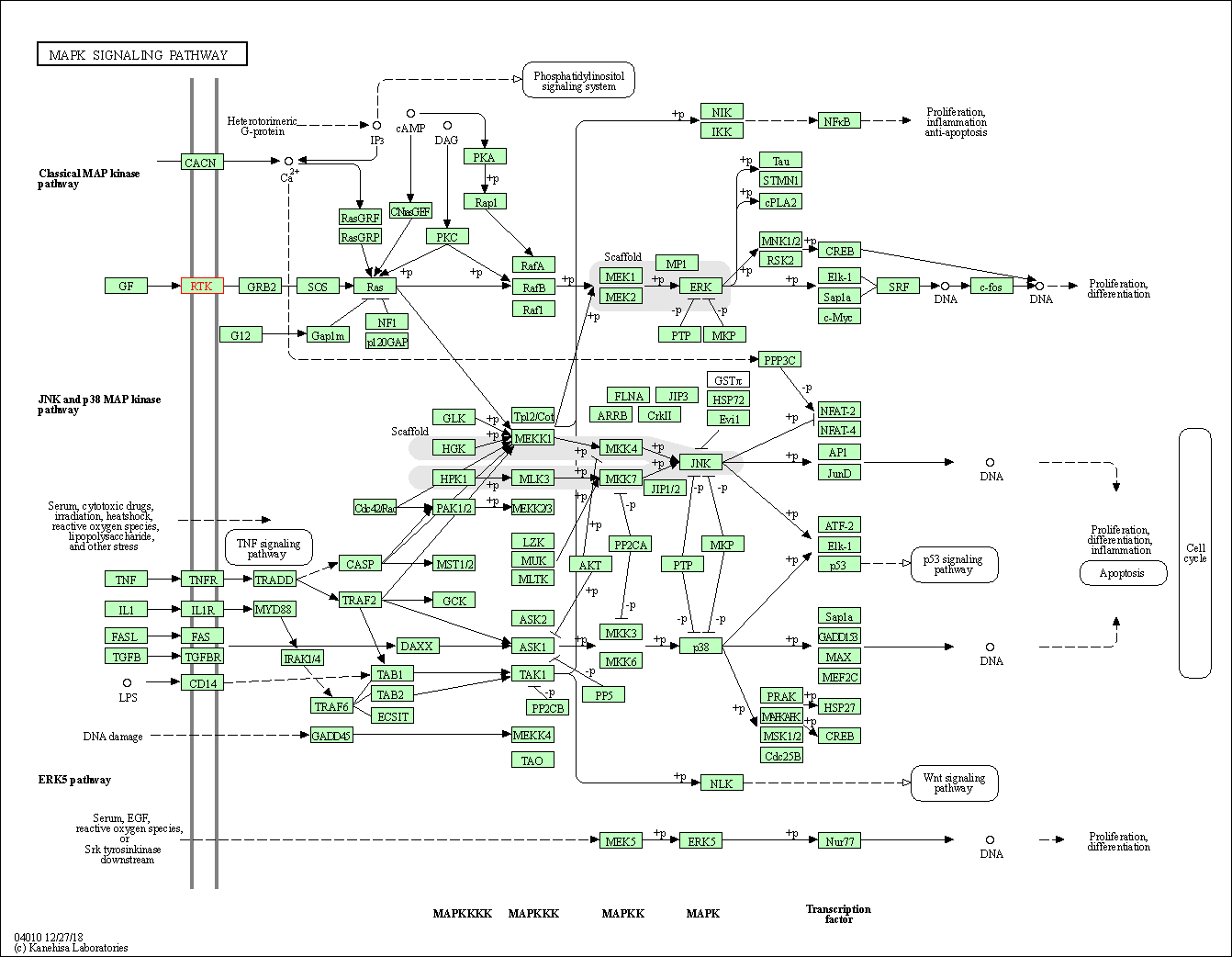
| Down-regulation | Phosphorylation | JNK1 | Molecule Info |
Pathway MAP
|
||
| In-vitro Model | MV4-11 | CVCL_0064 | Childhood acute monocytic leukemia | Homo sapiens | ||
| MOLM-13 | CVCL_2119 | Adult acute myeloid leukemia | Homo sapiens | |||
| THP-1 | CVCL_0006 | Childhood acute monocytic leukemia | Homo sapiens | |||
| HL-60 | CVCL_0002 | Adult acute myeloid leukemia | Homo sapiens | |||
| In-vivo Model | Xenograft tumors were generated by injecting subcutaneously 1*107 MV4-11 cells in 100 uL of PBS on left flank in nude mice. | |||||
| Experimental
Result(s) |
Arsenic trioxide potentiates Gilteritinib-induced apoptosis in FLT3-ITD positive leukemic cells via IRE1a-JNK-mediated endoplasmic reticulum stress. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Fms-like tyrosine kinase 3 (FLT-3) | Molecule Info | [1] | |
| Tyrosine-protein kinase UFO (AXL) | Molecule Info | [3] | ||
| KEGG Pathway | Cytokine-cytokine receptor interaction | Click to Show/Hide | ||
| 2 | Hematopoietic cell lineage | |||
| 3 | Pathways in cancer | |||
| 4 | Transcriptional misregulation in cancer | |||
| 5 | Acute myeloid leukemia | |||
| 6 | Central carbon metabolism in cancer | |||
| NetPath Pathway | FSH Signaling Pathway | Click to Show/Hide | ||
| Pathway Interaction Database | Validated transcriptional targets of deltaNp63 isoforms | Click to Show/Hide | ||
| Reactome | VEGFA-VEGFR2 Pathway | Click to Show/Hide | ||
| WikiPathways | miR-targeted genes in squamous cell - TarBase | Click to Show/Hide | ||
| 2 | miR-targeted genes in muscle cell - TarBase | |||
| 3 | miR-targeted genes in lymphocytes - TarBase | |||