Drug Details
| General Information of the Drug (ID: DR8384) | ||||
|---|---|---|---|---|
| Name |
Fluocinolone acetonide
|
|||
| Synonyms |
FLUOCINOLONE ACETONIDE; 67-73-2; Synalar; Flucinar; Synandone; Fluonid; Jellin; Synamol; Synemol; Coriphate; Fluovitif; Flupollon; Percutina; Synandrone; Dermalar; Localyn; Omniderm; Radiocin; Sinalar; Synotic; Tefunote; Synsac; Fluotrex; Retisert; Synalar-HP; Fluocet; Localyn Syntex; Derma-smoothe/fs; Cortiplastol; Prodermin; Fluocinolonacetonidum; Flucort; Fluocinolone 16,17-acetonide; Iluvien; Capex; 6alpha-Fluorotriamcinolone acetonide; Fluocinolone acetonide [DCIT]; Fluocinoloni acetonidum; RS-1401 AT; Fluocinoloni acetonidum [INN-Latin]; Acetonide de fluocinolone; C24H30F2O6; component of Neo-Synalar; Fluocinolone (Acetonide); UNII-0CD5FD6S2M; Dermatin (steroid); 6alpha,9alpha-Difluoro-16alpha-hydroxyprednisolone 16,17-acetonide; MFCD00010525; NSC-92339; 0CD5FD6S2M; MLS000028545; CHEBI:31623; Fluocinolone acetonide (Flucort-N); Fs Shampoo; NSC92339; flucinolone; SMR000058329; DSSTox_CID_20674; DSSTox_RID_79533; DSSTox_GSID_40674; Acetonida de fluocinolona; RS 1401AT; Flucort-N; 6.alpha.-Fluorotriamcinolone acetonide; CAS-67-73-2; Retisert (TN); (1S,2S,4R,8S,9S,11S,12R,13S,19S)-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-6,6,9,13-tetramethyl-5,7-dioxapentacyclo[10.8.0.02,9.04,8.013,18]icosa-14,17-dien-16-one; (6alpha,11beta,16alpha)-6,9-Difluoro-11,21-dihydroxy-16,17-[(1-methylethylidene)bis(oxy)]pregna-1,4-diene-3,20-dione; Fluocet (TN); Synalar (TN); Acetonide de fluocinolone [INN-French]; Acetonida de fluocinolona [INN-Spanish]; CCRIS 3250; HSDB 3083; 6.Alpha.,9-Difluoro; EINECS 200-668-5; NSC 92339; Dermatin; Fluzon [Steroid]; NCGC00021301-04; 6-alpha,9-alpha-Difluoro-16-alpha-hydroxyprednisolone 16,17-acetonide; Fluocinoloneacetonide; fluocinolone-acetonide; 6alpha,9-Difluoro-11beta,16alpha,17,21-tetrahydroxypregna-1,4-diene-3,20-dione, cyclic 16,17-acetal with acetone; Pregna-1,4-diene-3,20-dione, 6,9-difluoro-11,21-dihydroxy-16,17-((1-methylethylidene)bis(oxy))-, (6alpha,11beta,16alpha)-; Pregna-1,4-diene-3,20-dione, 6alpha,9-difluoro-11beta,16alpha,17,21-tetrahydroxy-, cyclic 16,17-acetal with acetone; Fluocinolone acetonide intravitreal implant; acetonido de fluocinolona; 6.alpha.,17-acetonide; Opera_ID_1295; F0657; Fluocinolone acetonide [USAN:USP:INN:JAN]; CHEMBL989; SCHEMBL4795; 6alpha,9alpha-Difluoro-11beta,16alpha,17alpha,21-tetrahydroxy-1,4-pregnadiene-3,20-dione 16,17-Acetonide; MLS001076276; ARONIS24561; GTPL7077; DTXSID0040674; EBD4852; HMS2090I14; HMS2230D13; HMS3259G12; HMS3715J11; HY-B0415; ZINC3977981; Tox21_110869; Tox21_302364; AC-429; s2470; AKOS015963144; Tox21_110869_1; ACN-043362; CCG-221165; DB00591; NC00562; Fluocinolone acetonide (JP17/USP/INN); NCGC00021301-06; NCGC00255654-01; (4aS,4bR,5S,6aS,6bS,9aR,10aS,10bS,12S)-4b,12-difluoro-5-hydroxy-6b-(hydroxyacetyl)-4a,6a,8,8-tetramethyl-4a,4b,5,6,6a,6b,9a,10,10a,10b,11,12-dodecahydro-2H-naphtho[2',1':4,5]indeno[1,2-d][1,3]dioxol-2-one; (4aS,4bR,5S,6aS,6bS,9aR,10aS,10bS,12S)-4b,12-difluoro-6b-glycoloyl-5-hydroxy-4a,6a,8,8-tetramethyl-4a,4b,5,6,6a,6b,9a,10,10a,10b,11,12-dodecahydro-2H-naphtho[2',1':4,5]indeno[1,2-d][1,3]dioxol-2-one; 4b,12-Difluoro-6b-glycoloyl-5-hydroxy-4a,6a,8,8-tetramethyl-4a,4b,5,6,6a,6b,9a,10,10a,10b,11,12-dodecahydro-2H-naphtho[2',1':4,5]indeno[1,2-d][1,3]dioxol-2-one; AS-13690; NCI60_042042; Pregna-1,4-diene-3,20-dione, 6alpha,9-difluoro-11beta,16alpha,17,21-tetrahydroxy-, cyclic 16,17-acetal with acetone (8CI); Fluocinolone acetonide, analytical standard; D01825; J10113; AB00383017-10; AB00383017_11; 010F525; Q924467; SR-01000000109; SR-01000000109-2; W-104704; BRD-K94353609-001-21-6; difluoro-hydroxy-(2-hydroxyacetyl)-tetramethyl-[?]one; 6.alpha., 9.alpha.-Difluoro-16.alpha.-hydroxyprednisolone 16,17-acetonide; Fluocinolone acetonide, European Pharmacopoeia (EP) Reference Standard; Fluocinolone acetonide, United States Pharmacopeia (USP) Reference Standard; Fluocinolone acetonide for system suitability, European Pharmacopoeia (EP) Reference Standard; Fluocinolone Acetonide, Pharmaceutical Secondary Standard; Certified Reference Material; WLN: T F5 E5 B666 GO IO RV AHTTTT&J A BF CQ E FVIQ H H OF -A&BHO -B&ACEF; (1S,2S,4R,8S,9S,11S,12R,13S,19S)-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-6,6,9,13-tetramethyl-5,7-dioxapentacyclo[10.8.0.0^{2,9}.0^{4,8}.0^{13,18}]icosa-14,17-dien-16-one; (2S,6aS,6bR,7S,8aS,8bS,11aR,12aS,12bS)-2,6b-difluoro-7-hydroxy-8b-(2-hydroxyacetyl)-6a,8a,10,10-tetramethyl-6a,6b,7,8,8a,8b,11a,12,12a,12b-decahydro-1H-naphtho[2',1':4,5]indeno[1,2-d][1,3]dioxol-4(2H)-one; 6.alpha.,16.alpha.,17,21-tetrahydroxypregna-1,4-diene-3,20-dione, cyclic 16,17-acetal with acetone; 6alpha,9alpha-difluoro-11beta,21-dihydroxy-16alpha-17alpha-isopropylidenedioxypregna-1,4-diene-3,20-dione;; Fluocinolone acetonide for peak identification, European Pharmacopoeia (EP) Reference Standard; Pregna-1,20-dione, 6,9-difluoro-11,12-dihydroxy-16,17-[(1-methylethylidene)bis(oxy)]-, (6.alpha.,11.beta.,16.alpha.)-; Pregna-1,20-dione, 6,9-difluoro-11,21-dihydroxy-16,17-[(1-methylethylidene)bis(oxy)-, (6.alpha.,11.beta.,16.alpha.)-; Pregna-1,20-dione, 6,9-difluoro-11,21-dihydroxy-16,17-[(1-methylethylidene)bis(oxy)]-, (6.alpha.,11.beta.,16.alpha.)-; Pregna-1,20-dione, 6.alpha.,9-difluoro-11.beta.,16.alpha.,17,21-tetrahydroxy-, cyclic 16,17-acetal with acetone
Click to Show/Hide
|
|||
| Molecular Type |
Small molecule
|
|||
| Disease | Inflammation [ICD-11: N.A.] | Approved | [1] | |
| Structure |
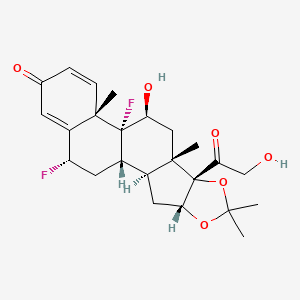
|
Click to Download Mol2D MOL |
||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | ||||
| Formula |
C24H30F2O6
|
|||
| PubChem CID | ||||
| Canonical SMILES |
CC1(OC2CC3C4CC(C5=CC(=O)C=CC5(C4(C(CC3(C2(O1)C(=O)CO)C)O)F)C)F)C
|
|||
| InChI |
1S/C24H30F2O6/c1-20(2)31-19-9-13-14-8-16(25)15-7-12(28)5-6-21(15,3)23(14,26)17(29)10-22(13,4)24(19,32-20)18(30)11-27/h5-7,13-14,16-17,19,27,29H,8-11H2,1-4H3/t13-,14-,16-,17-,19+,21-,22-,23-,24+/m0/s1
|
|||
| InChIKey |
FEBLZLNTKCEFIT-VSXGLTOVSA-N
|
|||
| CAS Number |
CAS 67-73-2
|
|||
| ChEBI ID | ||||
| TTD Drug ID | ||||
| DrugBank ID | ||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Natural Product(s) Able to Enhance the Efficacy of This Drug | ||||||
| Tretinoin | Homo sapiens | Click to Show/Hide the Molecular Data of This NP | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vivo Model | Clinical trial | |||||
| Experimental
Result(s) |
Combination therapy with hydroquinone, tretinoin, and fluocinolone is the most cost-effective treatment when compared with any of its dyads or with hydroquinone alone. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Glucocorticoid receptor (NR3C1) | Molecule Info | [3] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | Click to Show/Hide | ||
| NetPath Pathway | IL2 Signaling Pathway | Click to Show/Hide | ||
| 2 | TCR Signaling Pathway | |||
| Pathway Interaction Database | Regulation of nuclear SMAD2/3 signaling | Click to Show/Hide | ||
| 2 | Signaling events mediated by HDAC Class II | |||
| 3 | FOXA2 and FOXA3 transcription factor networks | |||
| 4 | Glucocorticoid receptor regulatory network | |||
| 5 | Regulation of Androgen receptor activity | |||
| 6 | AP-1 transcription factor network | |||
| Reactome | BMAL1:CLOCK,NPAS2 activates circadian gene expression | Click to Show/Hide | ||
| WikiPathways | Serotonin Receptor 4/6/7 and NR3C Signaling | Click to Show/Hide | ||
| 2 | SIDS Susceptibility Pathways | |||
| 3 | Nuclear Receptors Meta-Pathway | |||
| 4 | Endoderm Differentiation | |||
| 5 | Hair Follicle Development: Cytodifferentiation (Part 3 of 3) | |||
| 6 | Adipogenesis | |||
| 7 | Circadian Clock | |||
| 8 | Nuclear Receptors | |||

