Natural Product (NP) Details
| General Information of the NP (ID: NP5000) | |||||
|---|---|---|---|---|---|
| Name |
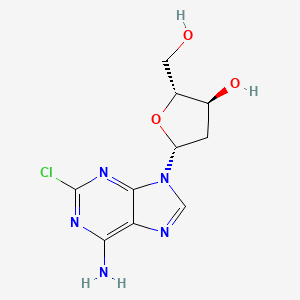
Cladribine
|
||||
| Synonyms |
Cladribine; 2-Chloro-2'-deoxyadenosine; 4291-63-8; Leustatin; 2-Chlorodeoxyadenosine; 2-CdA; Chlorodeoxyadenosine; CldAdo; ADENOSINE, 2-CHLORO-2'-DEOXY-; Litak; 2-Chloro-2'-deoxy-beta-adenosine; Cladaribine; RWJ 26251; 2-chloro-deoxyadenosine; 2ClAdo; UNII-47M74X9YT5; (2R,3S,5R)-5-(6-amino-2-chloro-9H-purin-9-yl)-2-(hydroxymethyl)tetrahydrofuran-3-ol; MLS000028377; (2R,3S,5R)-5-(6-amino-2-chloropurin-9-yl)-2-(hydroxymethyl)oxolan-3-ol; Cladarabine; RWJ-26251; Leustat; SMR000058553; 2-chloro-6-amino-9-(2-deoxy-beta-D-erythro-pentofuranosyl)purine; CHEBI:567361; 47M74X9YT5; MFCD00153939; NSC-105014; 2CdA; Movectro; Mylinax; DSSTox_CID_2828; (2R,3S,5R)-5-(6-amino-2-chloro-9H-purin-9-yl)-2-(hydroxymethyl)oxolan-3-ol; DSSTox_RID_76747; DSSTox_GSID_22828; cladribina; cladribinum; mavenclad; Leustatin (TN); CL9; CAS-4291-63-8; 2 Chlorodeoxyadenosine; SR-01000003063; NSC 105014; BRN 0624220; Cladiribine; CCRIS 9374; Adenosine, 2-chloro-2'-deoxy; HSDB 7564; 2-Chloro-6-amino-9-(2-deoxy-beta-D-erythropentofuranosyl)purine; Cladribine [USAN:USP:INN:BAN]; Mavenclad (TN); NCGC00018167-03; S1199; RWJ-26251-000; Opera_ID_1191; SCHEMBL3775; CHEMBL1619; Cladribine (JAN/USP/INN); 2-chloro-2'-deoxy-adenosine; cid_20279; MLS000028484; MLS000759397; MLS001077345; MLS001424194; GTPL4799; DTXSID8022828; BDBM38920; HMS2052K13; HMS2232C23; HMS3715F17; ACT02615; AMY22140; BCP02868; ZINC3798064; Tox21_110834; Tox21_300596; CC0168; NSC-05014; NSC-105014-F; AKOS015854898; AKOS015892544; AC-7591; BCP9000538; CCG-101116; CS-2057; DB00242; NC00366; NCGC00022567-05; NCGC00022567-06; NCGC00022567-07; NCGC00022567-08; NCGC00164384-01; NCGC00254518-01; 2-Chloro-2'-deoxyadenosine, antileukemic; AS-12366; BP-25407; HY-13599; AB0011806; SW197746-4; D01370; S-7479; AB00382963-17; AB00382963_19; 291C638; A826062; Q414030; SR-01000003063-7; SR-01000003063-10; Cladribine, European Pharmacopoeia (EP) Reference Standard; 6-amino-2-chloro-9-(2-deoxy-beta-erythropentofuranosyl)purine; Cladribine, United States Pharmacopeia (USP) Reference Standard; 6-amino-2-chloro-9-(2-deoxy-beta-D-erythro-pentofuranosyl)purine; (2R,3S,5R)-5-(6-amino-2-chloro-9-purinyl)-2-(hydroxymethyl)-3-oxolanol; (2R,3S,5R)-5-(6-Amino-2-chloropurin-9-yl)-2-(hydroxymethyl)oxalan-3-ol; (2R,3S,5R)-5-(6-amino-2-chloro-purin-9-yl)-2-(hydroxymethyl)tetrahydrofuran-3-ol; (2R,3S,5R)-5-(6-azanyl-2-chloranyl-purin-9-yl)-2-(hydroxymethyl)oxolan-3-ol; Cladribine for peak identification, European Pharmacopoeia (EP) Reference Standard; 24757-90-2; Leustatin; ; ; 2-Chloro-2'-deoxyadenosine; ; ; 2-CdA; ; ; NSC-105014-F; ; ; (2R,3S,5R)-5-(6-Amino-2-chloropurin-9-yl)-2-(hydroxymethyl)oxolan-3-ol
Click to Show/Hide
|
||||
| Species Origin | Homo sapiens ... | Click to Show/Hide | |||
| Homo sapiens | |||||
| Disease | Chronic lymphocytic leukaemia [ICD-11: 2A82] | Approved | [1] | ||
| Structure |
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-5.807
MDCK Permeability
-5.099
PAMPA
+++
HIA
- - -
Distribution
VDss
0.964
PPB
19.3%
BBB
-
Metabolism
CYP1A2 inhibitor
- - -
CYP1A2 substrate
- - -
CYP2C19 inhibitor
- - -
CYP2C19 substrate
- - -
CYP2C9 inhibitor
- - -
CYP2C9 substrate
- - -
CYP2D6 inhibitor
- - -
CYP2D6 substrate
- - -
CYP3A4 inhibitor
- - -
CYP3A4 substrate
- -
CYP2B6 inhibitor
++
CYP2B6 substrate
- - -
CYP2C8 inhibitor
- - -
HLM Stability
- - -
Excretion
CLplasma
12.562
T1/2
1.721
Toxicity
DILI
+++
Rat Oral Acute Toxicity
-
FDAMDD
+
Respiratory
-
Human Hepatotoxicity
++
Ototoxicity
++
Drug-induced Nephrotoxicity
++
Drug-induced Neurotoxicity
+++
Hematotoxicity
+
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C10H12ClN5O3
|
||||
| PubChem CID | |||||
| Canonical SMILES |
C1C(C(OC1N2C=NC3=C(N=C(N=C32)Cl)N)CO)O
|
||||
| InChI |
1S/C10H12ClN5O3/c11-10-14-8(12)7-9(15-10)16(3-13-7)6-1-4(18)5(2-17)19-6/h3-6,17-18H,1-2H2,(H2,12,14,15)/t4-,5+,6+/m0/s1
|
||||
| InChIKey |
PTOAARAWEBMLNO-KVQBGUIXSA-N
|
||||
| CAS Number |
CAS 4291-63-8
|
||||
| ChEBI ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Entinostat | Breast cancer | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
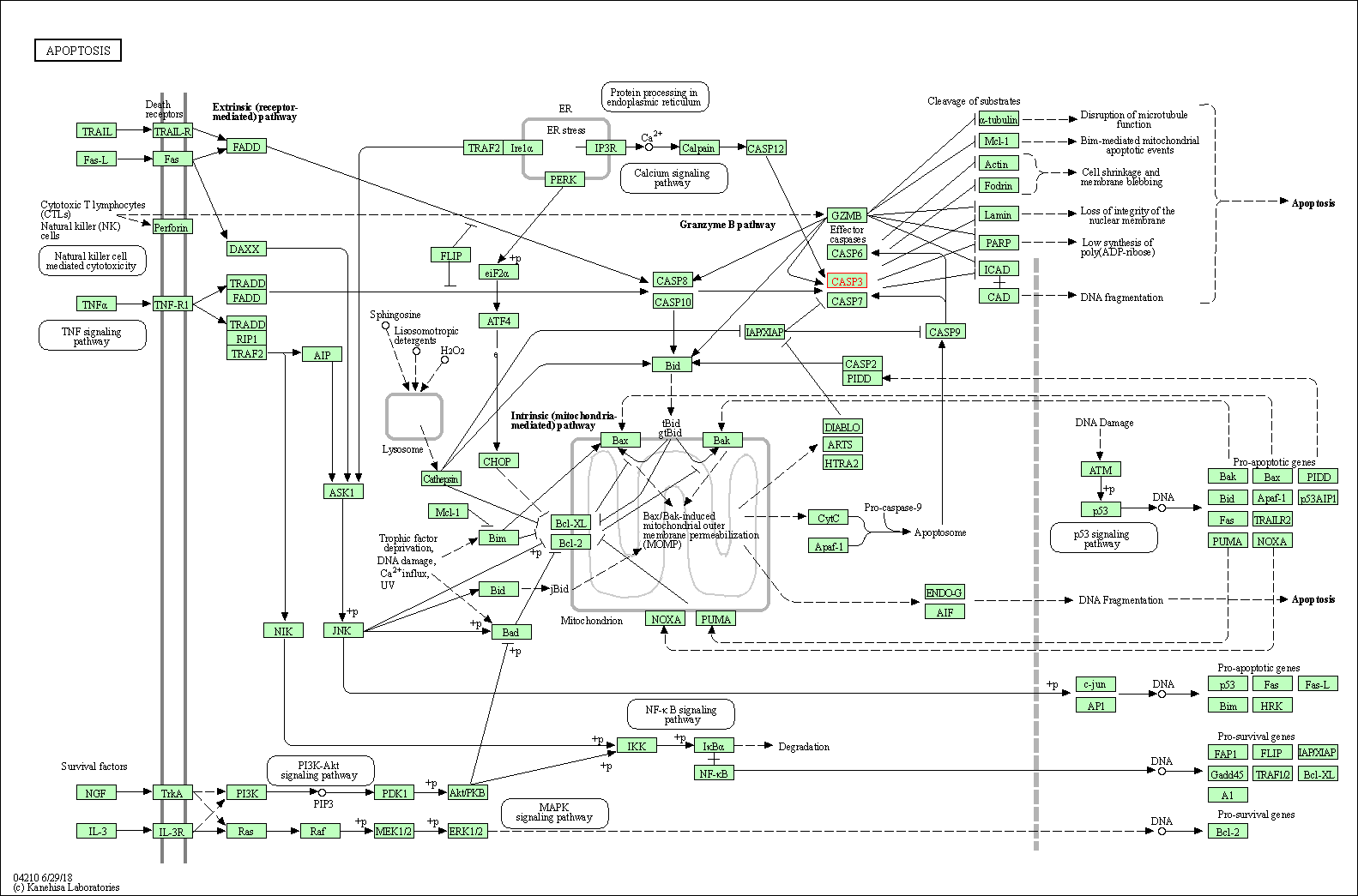
Up-regulation | Cleavage | CASP3 | Molecule Info |
Pathway MAP
|
|
| Up-regulation | Cleavage | CASP8 | Molecule Info |
Pathway MAP
|
||
| Up-regulation | Cleavage | CASP9 | Molecule Info |
Pathway MAP
|
||
| In-vitro Model | RPMI-8226 | CVCL_0014 | Plasma cell myeloma | Homo sapiens | ||
| U266B1 | CVCL_0566 | Plasma cell myeloma | Homo sapiens | |||
| MM1.R | CVCL_8794 | Plasma cell myeloma | Homo sapiens | |||
| Experimental
Result(s) |
Combinations of cladribine and entinostat exhibit potent activity to induce anti-proliferative/anti-survival effects on MM cells via induction of cell cycle G1 arrest, apoptosis, and DNA damage response. | |||||
| S3I-201 | Lupus nephritis | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [3] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
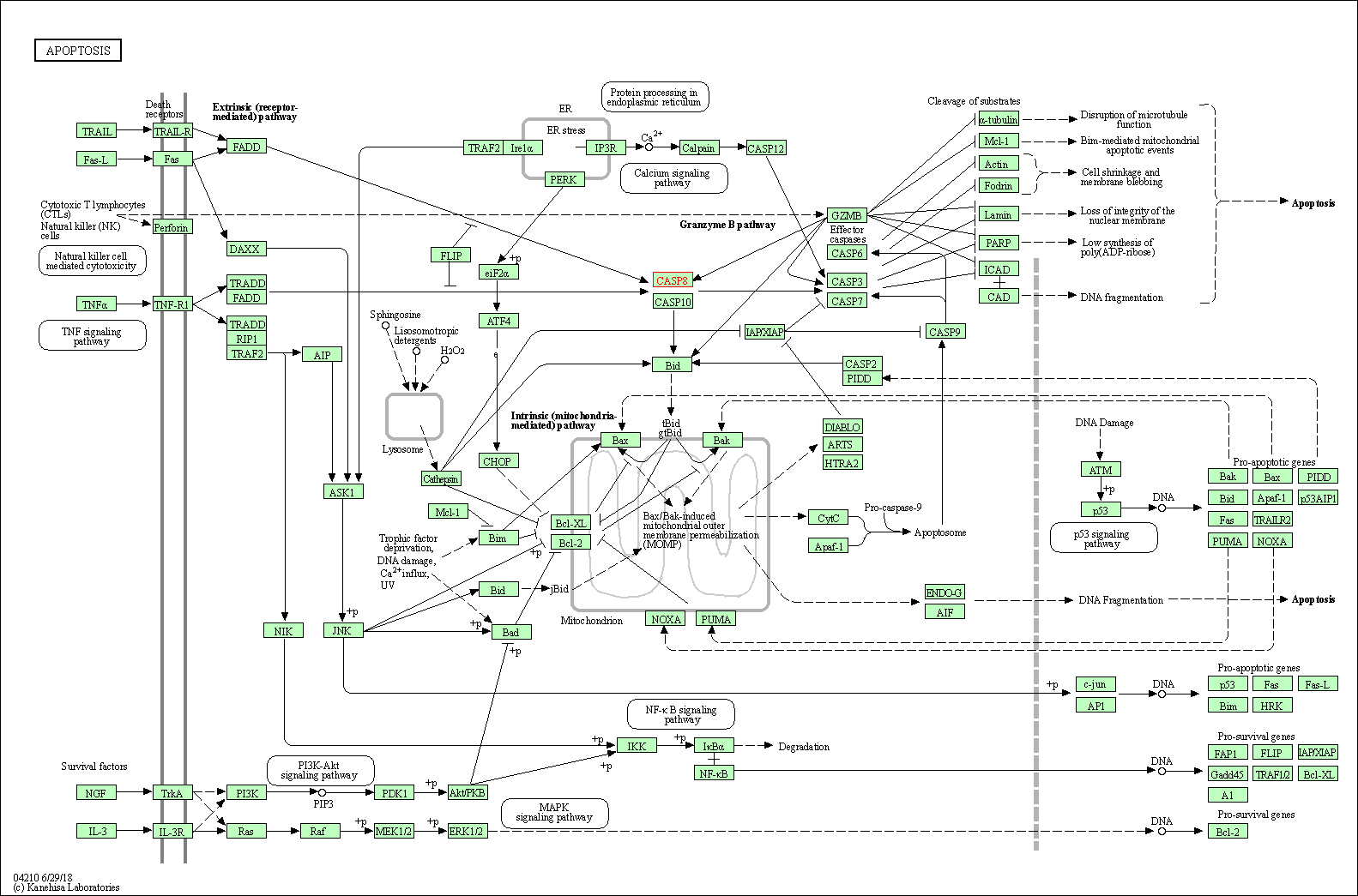
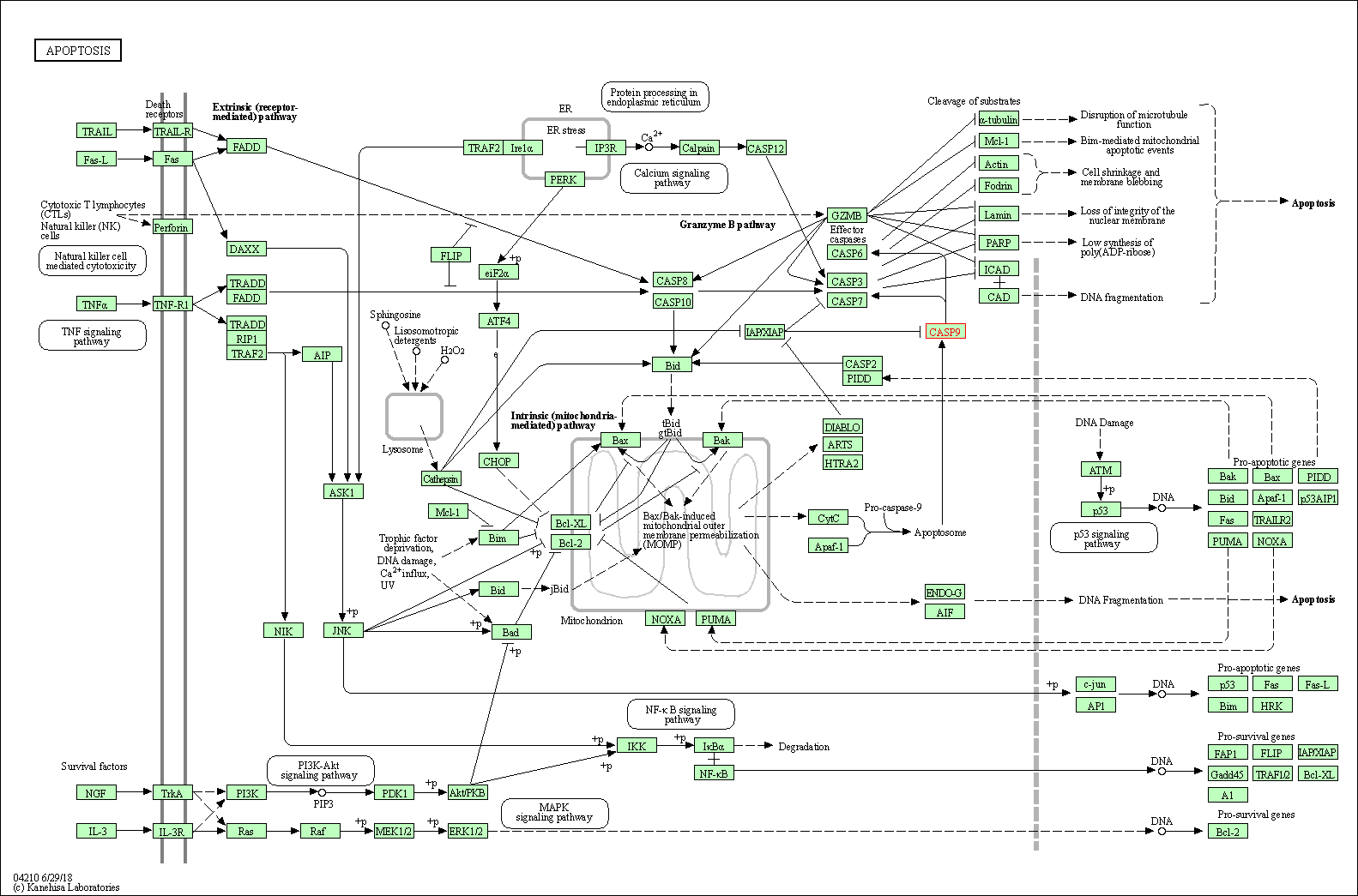
Up-regulation | Cleavage | CASP3 | Molecule Info |
Pathway MAP
|
|
| Up-regulation | Cleavage | CASP8 | Molecule Info |
Pathway MAP
|
||
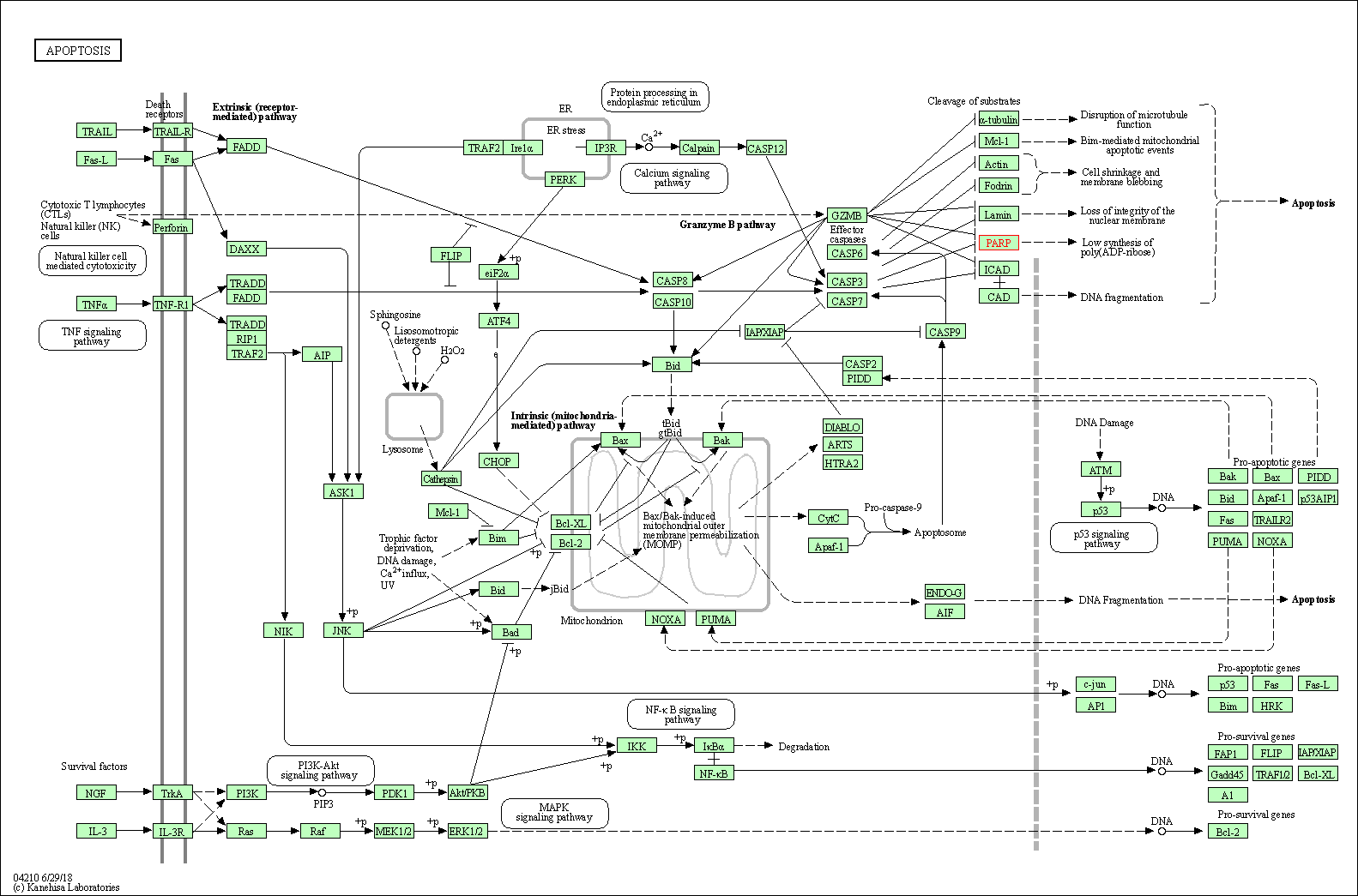
| Up-regulation | Cleavage | PARP1 | Molecule Info |
Pathway MAP
|
||
| In-vitro Model | U266B1 | CVCL_0566 | Plasma cell myeloma | Homo sapiens | ||
| RPMI-8226 | CVCL_0014 | Plasma cell myeloma | Homo sapiens | |||
| MM1.S | CVCL_8792 | Plasma cell myeloma | Homo sapiens | |||
| Experimental
Result(s) |
Cladribine exhibited inhibitory effects on MM cells in vitro. MM1.S is the only cell line showing significant response to the clinically achievable concentrations of cladribine-induced apoptosis and inactivation of STAT3. | |||||
| Rituximab | Malignant haematopoietic neoplasm | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Experimental
Result(s) |
The combination of a purine analog with rituximab was safe and effective for patients with recurrent HCL. | |||||
| Gemcitabine + Busulfan + Vorinostat | Click to Show/Hide the Molecular Data of This Drug | |||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [5] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | J45.01 | CVCL_2530 | T acute lymphoblastic leukemia | Homo sapiens | ||
| U-937 | CVCL_0007 | Adult acute monocytic leukemia | Homo sapiens | |||
| Experimental
Result(s) |
The Clad+Gem+Bu+SAHA combination provides synergistic cytotoxicity in lymphoma cell lines. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Adenosine deaminase (ADA) | Molecule Info | [6] | |
| BioCyc | Purine nucleotides degradation | Click to Show/Hide | ||
| 2 | Purine deoxyribonucleosides degradation | |||
| 3 | Purine ribonucleosides degradation to ribose-1-phosphate | |||
| 4 | Adenosine nucleotides degradation | |||
| 5 | Superpathway of purine nucleotide salvage | |||
| 6 | Adenine and adenosine salvage III | |||
| KEGG Pathway | Purine metabolism | Click to Show/Hide | ||
| 2 | Metabolic pathways | |||
| 3 | Primary immunodeficiency | |||
| NetPath Pathway | TCR Signaling Pathway | Click to Show/Hide | ||
| 2 | IL2 Signaling Pathway | |||
| Panther Pathway | Adenine and hypoxanthine salvage pathway | Click to Show/Hide | ||
| Pathwhiz Pathway | Purine Metabolism | Click to Show/Hide | ||
| Pathway Interaction Database | p73 transcription factor network | Click to Show/Hide | ||
| 2 | C-MYB transcription factor network | |||
| 3 | Validated transcriptional targets of deltaNp63 isoforms | |||
| 4 | Validated transcriptional targets of TAp63 isoforms | |||
| Reactome | Purine salvage | Click to Show/Hide | ||
| WikiPathways | Metabolism of nucleotides | Click to Show/Hide | ||