Natural Product (NP) Details
| General Information of the NP (ID: NP9241) | |||||
|---|---|---|---|---|---|
| Name |
Amentoflavone
|
||||
| Synonyms |
Amentoflavone; 1617-53-4; Didemethyl-ginkgetin; 3',8''-Biapigenin; Amenthoflavone; UNII-9I1VC79L77; I3,II8-biapigenin; MLS000574827; CHEBI:2631; 9I1VC79L77; MFCD00017470; 8-[5-(5,7-dihydroxy-4-oxo-chromen-2-yl)-2-hydroxy-phenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl)chromen-4-one; SMR000156235; 4H-1-Benzopyran-4-one, 8-[5-(5,7-dihydroxy-4-oxo-4H-1-benzopyran-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl)-; 8-(5-(5,7-dihydroxy-4-oxo-4H-1-benzopyran-2-yl)-2-hydroxyphenyl)-5,7-dihydroxy-2-(4-hydroxyphenyl)-4H-1-benzopyran-4-one; 8-(5-(5,7-Dihydroxy-4-oxo-4H-chromen-2-yl)-2-hydroxyphenyl)-5,7-dihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-one; 8-[5-(5,7-dihydroxy-4-oxo-4H-chromen-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-one; Tridemethylsciadopitysin; SR-01000721725; 4H-1-Benzopyran-4-one, 8-(5-(5,7-dihydroxy-4-oxo-4H-1-benzopyran-2-yl)-2-hydroxyphenyl)-5,7-dihydroxy-2-(4-hydroxyphenyl)-; NSC 295677; BIDD:PXR0028; SCHEMBL312563; MEGxp0_000924; med.21724, Compound 138; DTXSID20167225; Amentoflavone, analytical standard; BDBM429466; HMS2228B12; HMS3343J17; HMS3885A08; BCP13255; HY-N0662; ZINC3984030; Amentoflavone, >=99.0% (HPLC); LMPK12040009; MFCD20275041; NSC295677; s3833; (4-hydroxyphenyl)-4H-chromen-4-one; AKOS015896819; CCG-269950; CS-4945; NSC-295677; 3',8-Bi[4',5,7-trihydroxyflavone]; NCGC00247542-01; 8-[5-(5,7-dihydroxy-4-oxochromen-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl)chromen-4-one; AK167966; BS-15502; DB-043499; FT-0622262; N2268; W1536; C10018; 617A534; A810291; 8-(5-(5,7-dihydroxy-4-oxo-4H-chromen-2-yl)-2; Q-100192; Q4742425; SR-01000721725-2; SR-01000721725-3; 4',5,7-Trihydroxyflavone(3'->8)-4',5,7-trihydroxyflavone; 4',4''',5,5'',7,7''-Hexahydroxy-3''',8-biflavone, 8CI; 5,5',7,7'-Tetrahydroxy-2,2'-bis(4-hydroxyphenyl)-[3,8'-bi-4H-1-benzopyran]-4,4'-dione; 4H-1-Benzopyran-4-one, 8-[5-(5,7-dihydroxy-4-oxo-4H-1-benzopyran-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl); 8-[5-(5,7-dihydroxy-4-oxo-1-benzopyran-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl)-1-benzopyran-4-one; 8-[5-(5,7-dihydroxy-4-oxochromen-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl)chromen-4-one.; 8-[5-[5,7-bis(oxidanyl)-4-oxidanylidene-chromen-2-yl]-2-oxidanyl-phenyl]-2-(4-hydroxyphenyl)-5,7-bis(oxidanyl)chromen-4-one
Click to Show/Hide
|
||||
| Species Origin | Gingko biloba ... | Click to Show/Hide | |||
| Gingko biloba | |||||
| Disease | Epilepsy/seizure [ICD-11: 8A60] | Investigative | [1] | ||
| Structure |
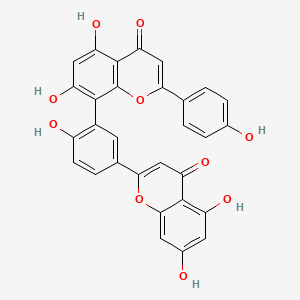
|
Click to Download Mol2D MOL |
|||
| ADMET Property |
Absporption
Caco-2 Permeability
-5.458
MDCK Permeability
-4.799
PAMPA
++
HIA
- - -
Distribution
VDss
-0.463
PPB
96.6%
BBB
- - -
Metabolism
CYP1A2 inhibitor
+++
CYP1A2 substrate
- -
CYP2C19 inhibitor
- - -
CYP2C19 substrate
- - -
CYP2C9 inhibitor
- - -
CYP2C9 substrate
- - -
CYP2D6 inhibitor
+++
CYP2D6 substrate
+++
CYP3A4 inhibitor
+++
CYP3A4 substrate
- - -
CYP2B6 inhibitor
- - -
CYP2B6 substrate
- - -
CYP2C8 inhibitor
+++
HLM Stability
+++
Excretion
CLplasma
1.96
T1/2
2.065
Toxicity
DILI
+++
Rat Oral Acute Toxicity
+
FDAMDD
+++
Respiratory
+++
Human Hepatotoxicity
-
Ototoxicity
- - -
Drug-induced Nephrotoxicity
- - -
Drug-induced Neurotoxicity
- - -
Hematotoxicity
- - -
Genotoxicity
+++
Tips: 1. For the classification endpoints, the prediction probability values are transformed into six symbols: 0-0.1 (- - -), 0.1-0.3 (- -), 0.3-0.5 (-), 0.5-0.7 (+), 0.7-0.9 (++), and 0.9-1.0 (+++).
2. Additionally, the corresponding relationships of the three labels are as follows: excellent; medium; poor.
Click to Show/Hide
|
||||
| Click to Show/Hide the Molecular Information and External Link(s) of This Natural Product | |||||
| Formula |
C30H18O10
|
||||
| PubChem CID | |||||
| Canonical SMILES |
C1=CC(=CC=C1C2=CC(=O)C3=C(O2)C(=C(C=C3O)O)C4=C(C=CC(=C4)C5=CC(=O)C6=C(C=C(C=C6O5)O)O)O)O
|
||||
| InChI |
1S/C30H18O10/c31-15-4-1-13(2-5-15)24-12-23(38)29-21(36)10-20(35)27(30(29)40-24)17-7-14(3-6-18(17)33)25-11-22(37)28-19(34)8-16(32)9-26(28)39-25/h1-12,31-36H
|
||||
| InChIKey |
YUSWMAULDXZHPY-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 1617-53-4
|
||||
| ChEBI ID | |||||
| Herb ID | |||||
| SymMap ID | |||||
| TCMSP ID | |||||
| TTD Drug ID | |||||
| Combinatorial Therapeutic Effect(s) Validated Clinically or Experimentally | ||||||
|---|---|---|---|---|---|---|
| α. A List of Drug(s) Whose Efficacy can be Enhanced by This NP | ||||||
| Cisplatin | Bladder cancer | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [2] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
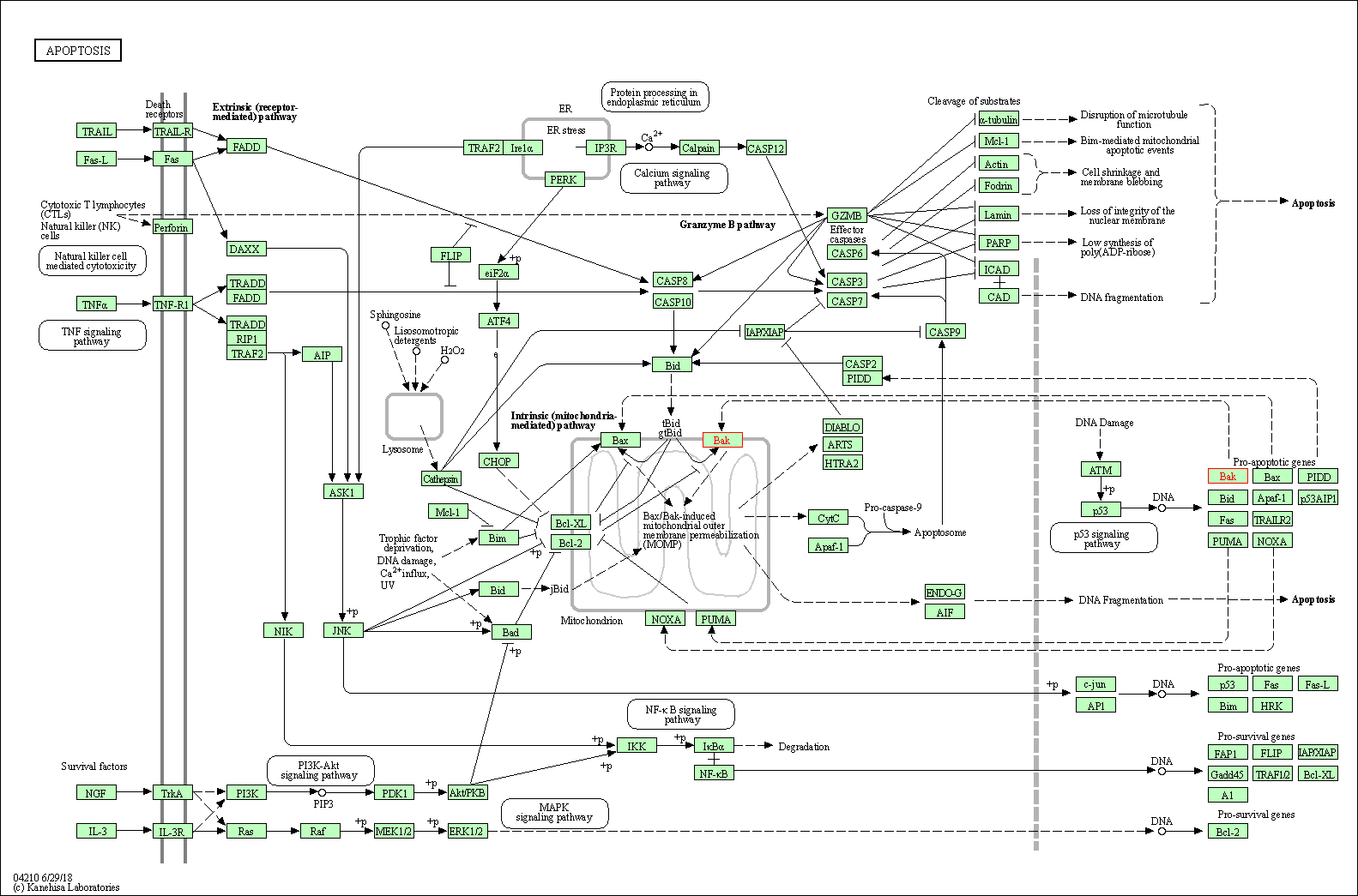
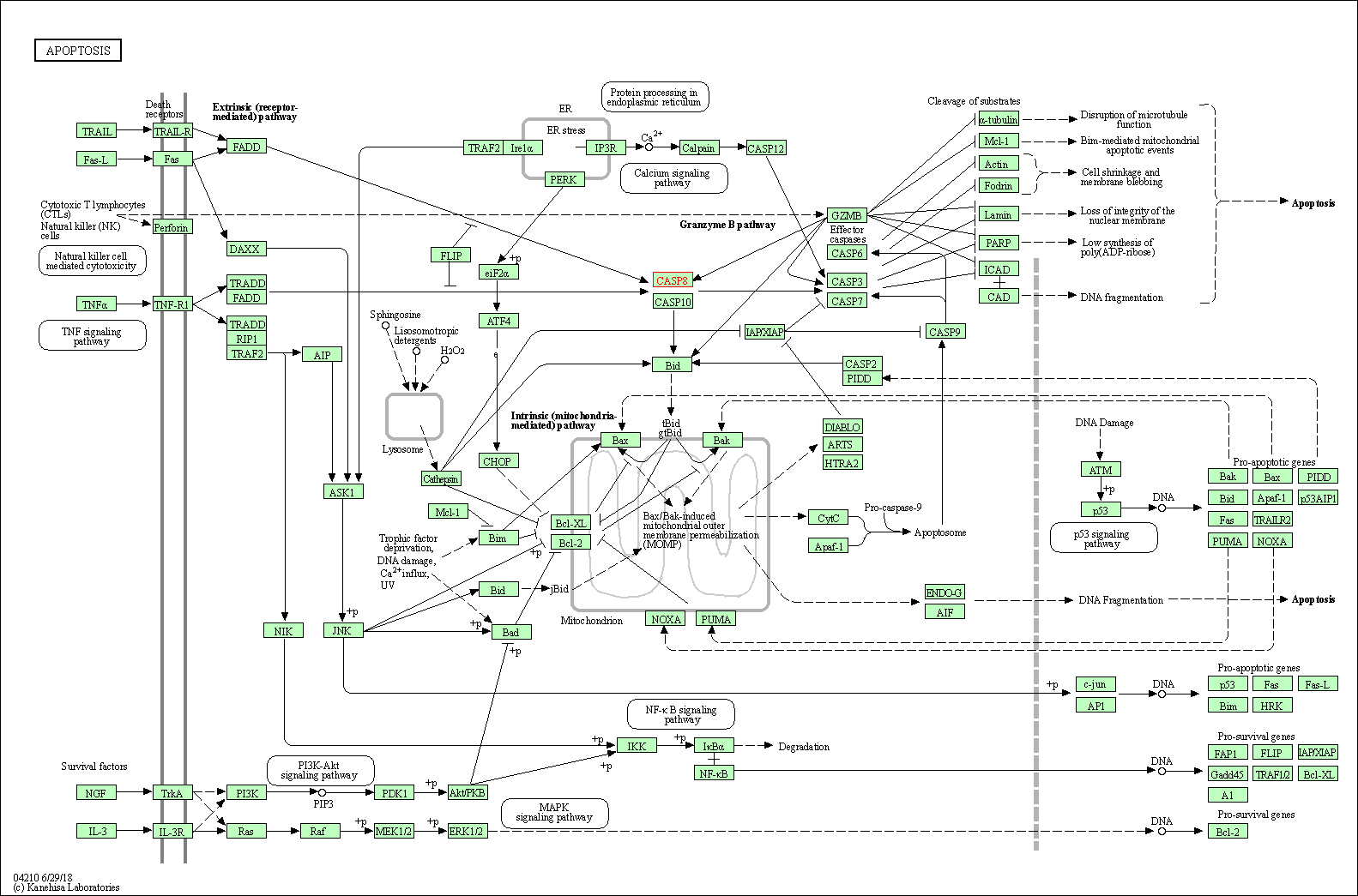
Up-regulation | Expression | BAK | Molecule Info |
Pathway MAP
|
|
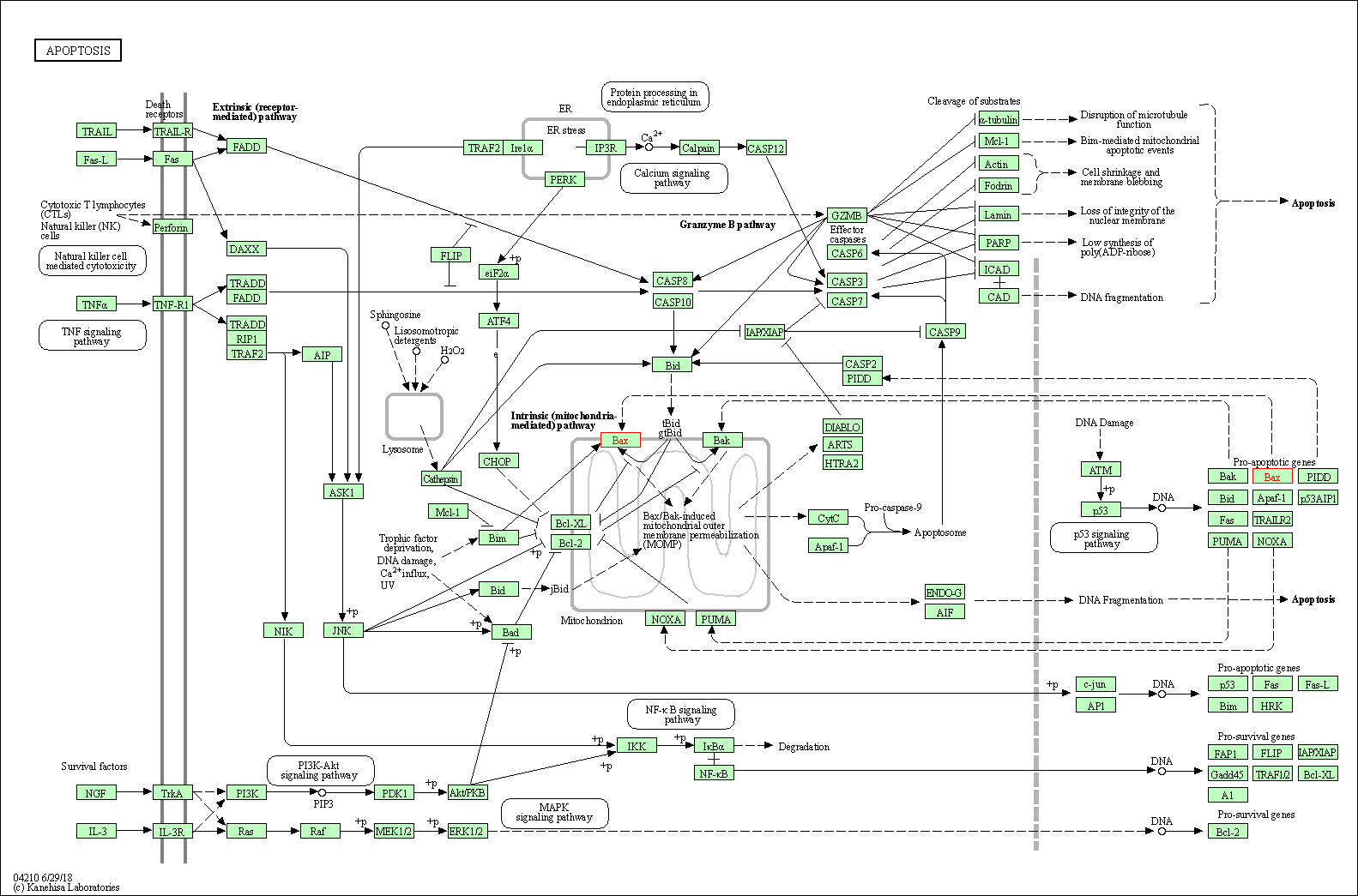
| Up-regulation | Expression | BAX | Molecule Info |
Pathway MAP
|
||
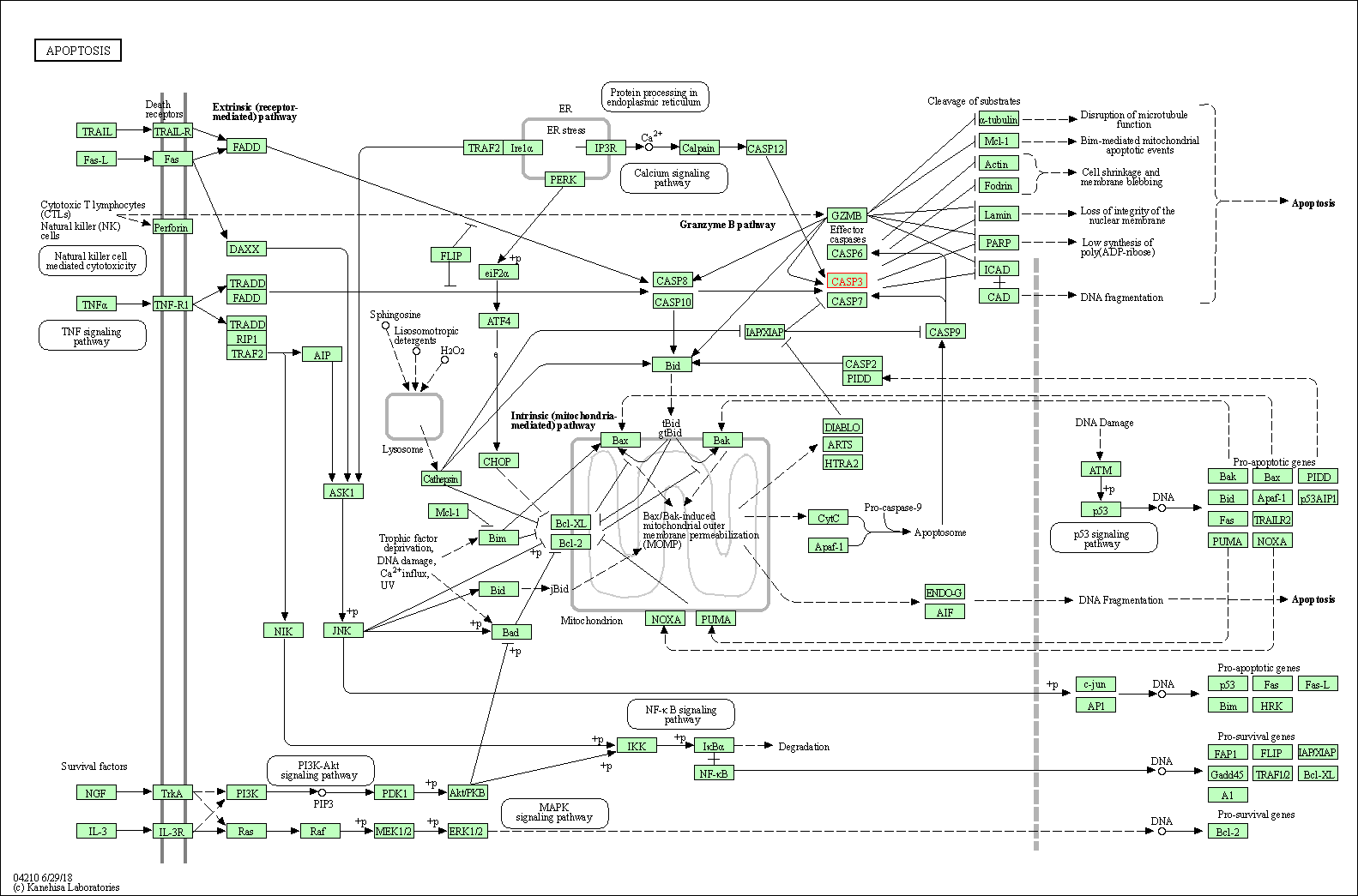
| Up-regulation | Cleavage | CASP3 | Molecule Info |
Pathway MAP
|
||
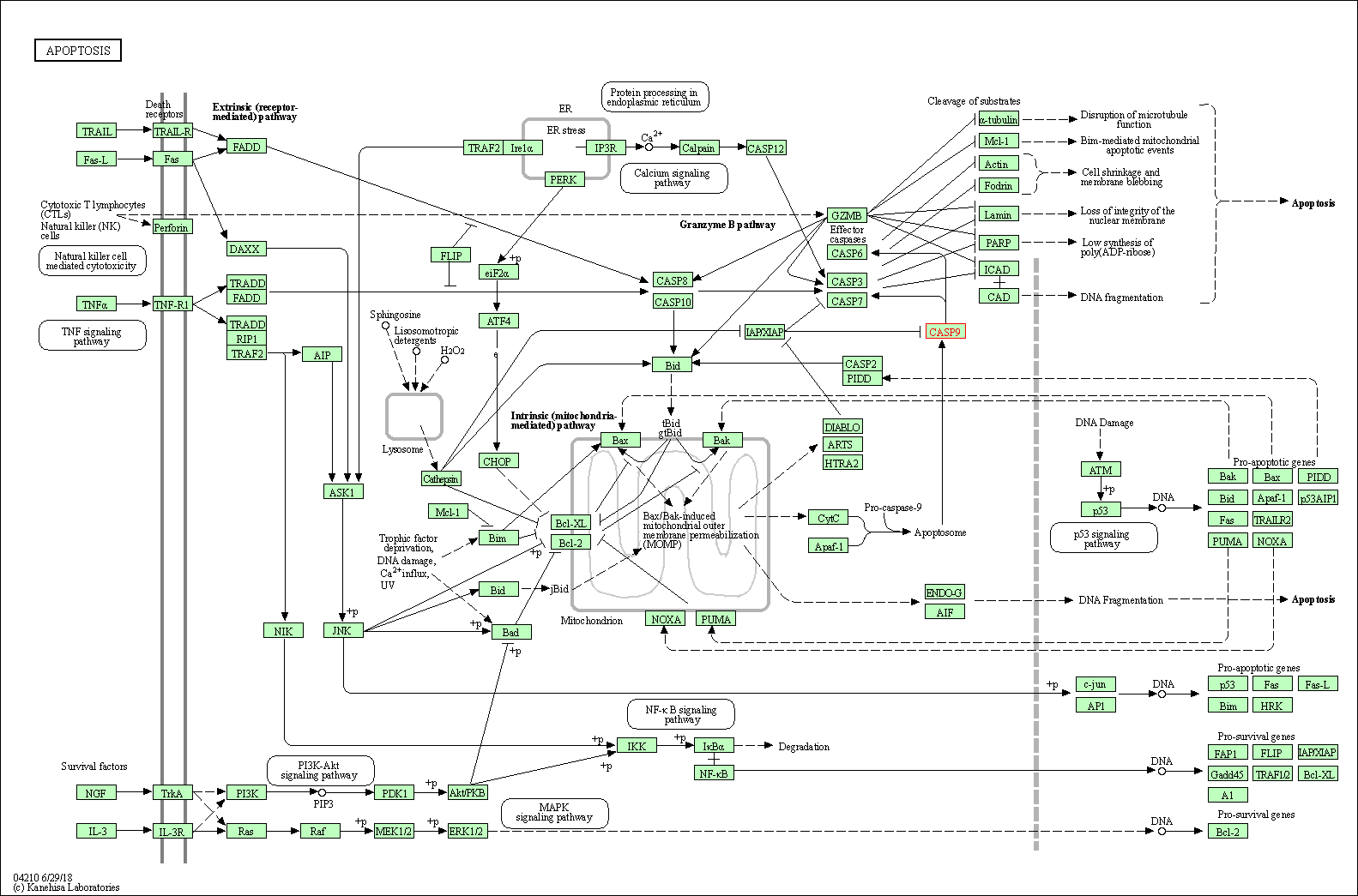
| Up-regulation | Cleavage | CASP9 | Molecule Info |
Pathway MAP
|
||
| In-vitro Model | SAS | CVCL_1675 | Tongue squamous cell carcinoma | Homo sapiens | ||
| Experimental
Result(s) |
Inactivation of NF-KappaB and induction of apoptosis through intrinsic caspase-dependent and independent apoptotic pathways are associated with amentoflavone enhanced anti-OSCC efficacy of cisplatin. | |||||
| Sorafenib | Renal cell carcinoma | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [3] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Down-regulation | Expression | CASP3 | Molecule Info |
Pathway MAP
|
|
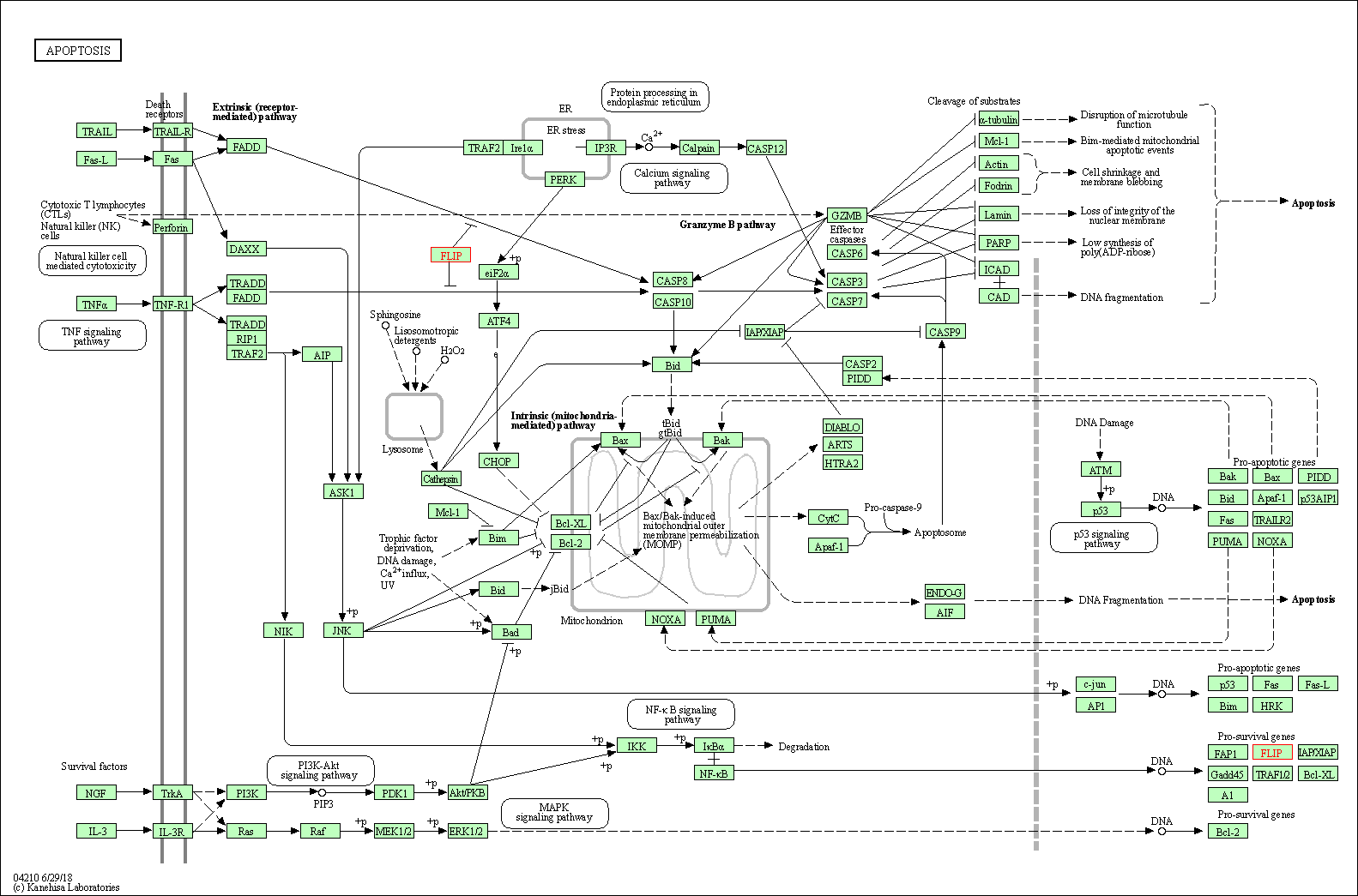
| Down-regulation | Expression | CASP8 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | CASP9 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | CFLAR | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Phosphorylation | ERK1 | Molecule Info |
Pathway MAP
|
||
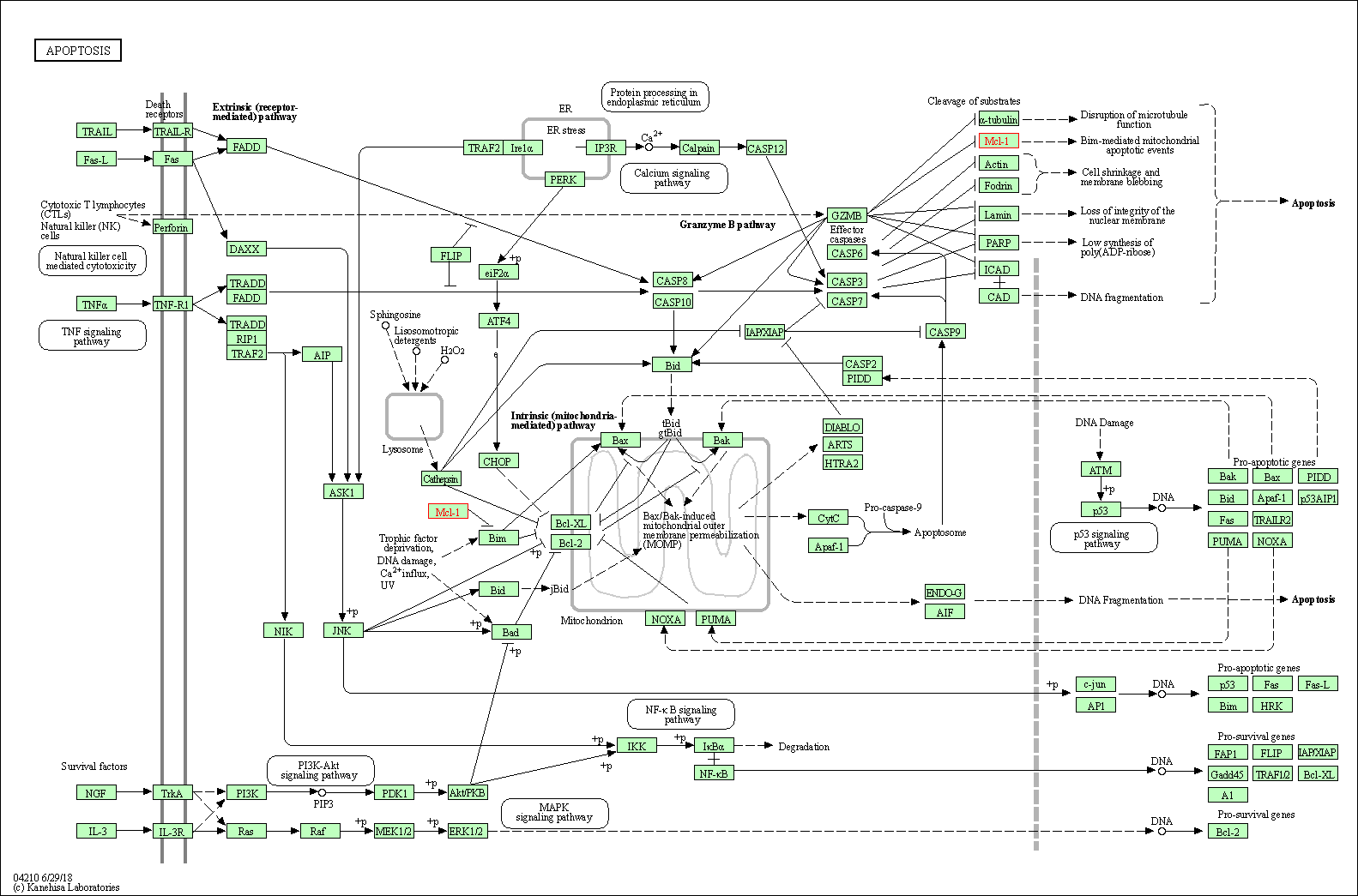
| Down-regulation | Expression | MCL1 | Molecule Info |
Pathway MAP
|
||
| Down-regulation | Expression | XIAP | Molecule Info |
Pathway MAP
|
||
| In-vivo Model | To establish the animal model, 1*107 SK-Hep1 cells were suspended in 150 ul mix-ture of serum-free DMEM and matrigel (2: 1) and inoculated subcutaneously in the right legs of nude mice. | |||||
| Experimental
Result(s) |
Amentoflavone boosts therapeutic efficacy of sorafenib through blockage of anti-apoptotic potential and induction of apoptosis in HCC in vivo. | |||||
| Ampicillin | Bacterial infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Enterococcus faecium ATCC 19434 | Microorganism model | Enterococcus faecium | |||
| Staphylococcus aureus ATCC 25923 | Microorganism model | Staphylococcus aureus | ||||
| Streptococcus mutans ATCC 3065 | Microorganism model | Streptococcus mutans | ||||
| Escherichia coli ATCC 43895 | Microorganism model | Escherichia coli | ||||
| Escherichia coli ATCC 25922 | Microorganism model | Escherichia coli | ||||
| Pseudomonas aeruginosa ATCC 27853 | Microorganism model | Pseudomonas aeruginosa | ||||
| Experimental
Result(s) |
Amentoflavone had a considerable antibacterial effect and synergistic interaction with antibiotics against various bacterial strains. The formation of hydroxyl radical would be a cause of the synergistic effect and that this oxidative stress originated from a transient NADH depletion. . | |||||
| Cefotaxime | Bacterial infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Enterococcus faecium ATCC 19434 | Microorganism model | Enterococcus faecium | |||
| Staphylococcus aureus ATCC 25923 | Microorganism model | Staphylococcus aureus | ||||
| Streptococcus mutans ATCC 3065 | Microorganism model | Streptococcus mutans | ||||
| Escherichia coli ATCC 43895 | Microorganism model | Escherichia coli | ||||
| Escherichia coli ATCC 25922 | Microorganism model | Escherichia coli | ||||
| Pseudomonas aeruginosa ATCC 27853 | Microorganism model | Pseudomonas aeruginosa | ||||
| Experimental
Result(s) |
Amentoflavone had a considerable antibacterial effect and synergistic interaction with antibiotics against various bacterial strains. The formation of hydroxyl radical would be a cause of the synergistic effect and that this oxidative stress originated from a transient NADH depletion. . | |||||
| Chloramphenicol | Bacterial infection | Click to Show/Hide the Molecular Data of This Drug | ||||
| Achieving Therapeutic Synergy | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [4] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| In-vitro Model | Enterococcus faecium ATCC 19434 | Microorganism model | Enterococcus faecium | |||
| Staphylococcus aureus ATCC 25923 | Microorganism model | Staphylococcus aureus | ||||
| Streptococcus mutans ATCC 3065 | Microorganism model | Streptococcus mutans | ||||
| Escherichia coli ATCC 43895 | Microorganism model | Escherichia coli | ||||
| Escherichia coli ATCC 25922 | Microorganism model | Escherichia coli | ||||
| Pseudomonas aeruginosa ATCC 27853 | Microorganism model | Pseudomonas aeruginosa | ||||
| Experimental
Result(s) |
Amentoflavone had a considerable antibacterial effect and synergistic interaction with antibiotics against various bacterial strains. The formation of hydroxyl radical would be a cause of the synergistic effect and that this oxidative stress originated from a transient NADH depletion. . | |||||
| β. A List of Drug(s) Whose Adverse Effect can be Decreased by This NP | ||||||
| Gamma-ray irradiation | Brain metastases | Click to Show/Hide the Molecular Data of This Drug | ||||
| Decreasing Adverse Drug Reaction | Click to Show/Hide | |||||
| Representative Experiment Reporting the Effect of This Combination | [5] | |||||
| Detail(s) |
Combination Info
 click to show the detail info of this combination
click to show the detail info of this combination
|
|||||
| Molecule(s)
Regulation |
Up-regulation | Expression | TNFAIP2 | Molecule Info | ||
| In-vivo Model | Male C57BL/6 mice(18-22g) were exposed to 60Co Gamma-irradiation at a dose of 7.5 Gy or 3 Gy and the dose rate was 1.58 Gy/min | |||||
| Experimental
Result(s) |
AMF is a potential protective agent against radiation injury. | |||||
| Target and Pathway | ||||
|---|---|---|---|---|
| Target(s) | Bcl-2-like protein 3 (MCL1) | Molecule Info | [6] | |
| Gamma-aminobutyric acid receptor (GAR) | Molecule Info | [7] | ||
| Xanthine dehydrogenase/oxidase (XDH) | Molecule Info | [8] | ||
| BioCyc | Purine nucleotides degradation | Click to Show/Hide | ||
| 2 | Urate biosynthesis/inosine 5'-phosphate degradation | |||
| 3 | Guanosine nucleotides degradation | |||
| 4 | Adenosine nucleotides degradation | |||
| 5 | Retinoate biosynthesis II | |||
| KEGG Pathway | PI3K-Akt signaling pathway | Click to Show/Hide | ||
| 2 | MicroRNAs in cancer | |||
| 3 | Purine metabolism | |||
| 4 | Caffeine metabolism | |||
| 5 | Drug metabolism - other enzymes | |||
| 6 | Metabolic pathways | |||
| 7 | Peroxisome | |||
| NetPath Pathway | TCR Signaling Pathway | Click to Show/Hide | ||
| Panther Pathway | Apoptosis signaling pathway | Click to Show/Hide | ||
| 2 | CCKR signaling map ST | |||
| 3 | Adenine and hypoxanthine salvage pathway | |||
| 4 | Purine metabolism | |||
| Pathwhiz Pathway | Caffeine Metabolism | Click to Show/Hide | ||
| 2 | Purine Metabolism | |||
| Pathway Interaction Database | E2F transcription factor network | Click to Show/Hide | ||
| 2 | Direct p53 effectors | |||
| 3 | IL6-mediated signaling events | |||
| 4 | HIF-1-alpha transcription factor network | |||
| Reactome | Purine catabolism | Click to Show/Hide | ||
| WikiPathways | Apoptosis | Click to Show/Hide | ||
| 2 | miR-targeted genes in muscle cell - TarBase | |||
| 3 | miR-targeted genes in lymphocytes - TarBase | |||
| 4 | miR-targeted genes in leukocytes - TarBase | |||
| 5 | Apoptosis Modulation and Signaling | |||
| 6 | Oxidative Stress | |||
| 7 | Effects of Nitric Oxide | |||
| 8 | Metabolism of nucleotides | |||
| 9 | Selenium Micronutrient Network | |||